Explore 2022 Research
The Healthcare Burden of Giant Cell Arteritis in South Australia
| Funded by: | Australian Rheumatology Association Research Fund |
| Recipient: | Dr Suellen Lyne |
| Intended Department | The Queen Elizabeth Hospital, Adelaide |
| Project: | The Healthcare Burden of Giant Cell Arteritis in South Australia |
Stratified care integrated with eHealth for low back pain
| Funded by: | Australian Rheumatology Association Research Fund |
| Recipient: | Dr Joshua Zadro |
| Intended Department | The University of Sydney, Institute for Musculoskeletal Health, Sydney School of Public Health, Faculty of Medicine and Health |
| Project: | Stratified care integrated with eHealth for low back pain |
Scientific summary:
What were the main scientific objectives of the grant?
To determine the feasibility of delivering stratified care integrated with eHealth (‘Rapid Virtual Stratified Care’) for patients with low back pain referred to an outpatient clinic and evaluating its effectiveness in a multi-centre randomised controlled trial.
To describe waiting times, number of appointments, intervention and healthcare costs, and the following clinical outcomes in the two arms of the trial (Rapid Virtual Stratified Care and usual care).
What were the main scientific achievements of the grant?
We recruited 40 participants from the Back Clinic to our pilot trial over 134 weeks, largely because the Back Clinic is only open for 4 hours of the week and we experience significant staffing issues post COVID. Adjusting for these reduced hours and disruptions, the recruitment rate for this pilot trial was approximately 3 participants per week. Of the 133 participants contacted to be part of the study, 40 provided consent (one withdrew due to medical issues); a consent rate of approximately 30%.
One of the main objectives of our pilot trial was to determine if this model of care could reduce wait times without compromising patient outcomes. We found that the median wait time to receive care was 29 weeks (IQR 7 to 89) in the usual care group and 16 weeks (IQR 7 to 30) in the intervention group. Patient outcomes were comparable between groups. For example, median pain scores at 6 weeks follow-up were 6 on a 0-10 scale (interquartile range, IQR: 4 to 7) in the usual care group and 7 (IQR 5 to 7) in the intervention group. This is particularly reassuring since those in the intervention group appeared to have more severe symptoms overall.
What problems, if any, did you encounter in achieving the project’s objectives, and how did you address them?
Recruitment was significantly delayed due to COVID-19 and the closure of the Back Clinic as resources and staff in the hospital were understandably relocated to other areas of need. This not only delayed our recruitment; however, resources and staff were unfortunately not re-allocated back to the clinic post COVID-19, which meant recruitment and implementation of the trial was slower than expected. These issues were addressed by extending our recruitment periods and use of research assistants to assist clinicians in the Back Clinic where possible (e.g., administrative tasks). This freed up time for clinicians to focus on the more urgent aspects of the trial and clinical practice.
Another major issue was appointment attendance rates and the ability to contact participants. Unfortunately, some patients did not attend their allocated face to face appointment or were unable to be contacted by the research assistant. We established processes to try and minimise this such as, reminder letters, reminder emails, and additional call back services at the expense of the research group. This unfortunately is not something that the current health-care system has the resources to implement.
What is the plan for dissemination of findings?
We have published the protocol at BMJ Open:
Zadro JR, Needs C, Foster NE, Martens D, Coombs DM, Machado GC, Adams C, Han CS, Maher CG. Feasibility of delivering and evaluating stratified care integrated with telehealth (‘Rapid Stratified Telehealth’) for patients with low back pain: protocol for a feasibility and pilot randomised controlled trial. BMJ Open. 2022 Jan 11;12(1):e056339. doi: 10.1136/bmjopen-2021-056339. PMID: 35017255; PMCID: PMC8753403.
We have presented preliminary results at the International Forum for Back and Neck Pain in Primary Care in August 2023, Groningen, Netherlands as a poster. We are yet to publish the final results in a scientific journal as we are completing the final stages of writing the manuscript.
Lay summary:
Overview:
We conducted a small-scale randomised controlled trial to investigate the feasibility of delivering and evaluating a new model of care for people with low back pain (‘Rapid Stratified Telehealth’) referred to a public hospital outpatient clinic. The model aims to reduce treatment waiting times without compromising clinical outcomes. This pilot study was the first step in a larger program of work that aims to develop a model of care that can reduce waiting times for people who seek care for musculoskeletal conditions in the public hospital system.
Overall, we recruited 40 patients with low back pain who were referred from their General Practitioner to the Back Clinic at Royal Prince Alfred Hospital; a hospital outpatient clinic where rheumatologists typically refer patients who would benefit from exercise and other physiotherapy-related interventions to physiotherapy. Our pilot trial highlighted the feasibility of delivering and evaluating our new model of care (Rapid Stratified Telehealth), with lower waiting times in the intervention group compared to those in the usual care group and similar patient outcomes.
We are now evaluating an adapted version of this model of care for a range of musculoskeletal conditions presenting to outpatient physiotherapy departments as part of a multisite clinical trial funded by a $1.5 million Medical Research Future Fund (MRFF) grant. The pilot trial funded by Arthritis Australia provided vital data on feasibility which informed this larger study.
What problems did you try to solve, or gaps in knowledge did you try to fill?
Low back pain is the leading cause of disability in Australia and globally. The length of waiting times is an important barrier to accessing recommended care for low back pain in the public system (e.g. advice to stay active, exercise), especially since 55% Australians do not have private health insurance. Long waiting times can delay recovery and lead to the development of chronic and disabling symptoms that become difficult to manage and require more intensive, costly treatment.
We developed a new model of care (Rapid Stratified Telehealth) that can identify simple cases suitable to be managed using less resources (e.g. telephone advice, App-based exercise program), thereby freeing-up clinic-based resources for those with more complex presentations.
Our model of care involved matching the type and amount of care patients receive to their risk of persistent symptoms, and was designed for patients with low back pain referred to a multidisciplinary rheumatology and physiotherapy clinic. As part of this model, participants at low risk of persistent symptoms were given simple advice and education over the telephone by a rheumatologist and were instructed to call back if their symptoms did not improve in 6 weeks. Participants at medium and high risk of persistent symptoms were offered a course of telehealth (video conference) physiotherapy targeting exercise-based self-management. High risk participants were additionally encouraged to complete an online self-directed pain education program. Participants with back pain that travelled down their leg were offered a clinic-based rheumatology appointment with the option of clinic-based physiotherapy.
The primary aim of our pilot randomised controlled trial was to determine the feasibility of i) delivering our new model of care (‘Rapid Stratified Telehealth’) for patients with low back pain; and ii) evaluating its effectiveness in a multi-centre randomised controlled trial. The secondary aims were to describe waiting times, clinical outcomes (pain, function, quality of life, satisfaction), healthcare use and adverse events in the two arms of the trial (Rapid Stratified Telehealth and usual care).
The overall objective of this program of work is to determine whether a new model of managing the waiting list (Rapid Stratified Telehealth) can reduce treatment waiting times while not compromising clinical outcomes compared to usual care for musculoskeletal conditions.
What did you discover during the course of the grant?
We recruited 40 participants from the Back Clinic to our pilot trial over 134 weeks, largely because the Back Clinic was only open for 4 hours of the week and we experienced significant staffing issues post COVID. Adjusting for these reduced hours and disruptions, the recruitment rate for this pilot trial was approximately 3 participants per week. Of the 133 participants contacted to be part of the study, 40 provided consent; a consent rate of approximately 30%. This highlights the feasibility of evaluating this model of care in a larger trial.
One of the main objectives of our study was to determine if this model of care could reduce wait times while not compromising clinical outcomes. Our pilot trial found that the median wait time to receive care was 29 days (IQR 7 to 89) in the usual care group and 16 weeks (IQR 7 to 30) in the intervention group. Patient outcomes were comparable between groups. For example, median pain scores at 6 weeks follow-up were 6 on a 0-10 scale (interquartile range, IQR: 4 to 7) in the usual care group and 7 (IQR 5 to 7) in the intervention group. This is particularly reassuring since those in the intervention group appeared to have more severe symptoms overall.
Given the potential benefit of our model of care on reducing wait times and data highlighting feasibility of conducting a larger trial, there is justification for an adequately powered randomised controlled trial to provide more definite evidence on the beneficial effects of our new model of care.
Have the findings of the research already benefited people with musculoskeletal disease? How might the findings inform further research to help sufferers in the future?
As part of the pilot trial, participants have already benefited from shorter wait times, allowing for faster and more efficient access to necessary healthcare services. Virtual care is also a more easily accessible mode of care for patients and less costly one for the healthcare system.
The model of care tested in this pilot trial has already informed a large, multisite trial investigating a similar model of care designed to apply to a range of musculoskeletal conditions that physiotherapists manage in public hospital outpatient clinics. This larger trial will provide definitive data on the effectiveness, cost-effectiveness and potential for implementation of this model of care, which could have substantial benefits for the millions of Australian’s who suffer from musculoskeletal conditions.
Are you planning to continue the research?
We are currently planning a randomised controlled trial evaluating the effectiveness of this new model of care based on the learnings of this pilot trial. This trial will include people with a range of musculoskeletal conditions, not only low back pain.
A combined program of education and exercise versus general advice for ankle osteoarthritis: A feasibility randomised controlled trial
| Funded by: | ARA Research Fund |
| Recipient: | Dr Bill Vicenzino |
| Intended Department | School of Health Sciences, College of Health, Medicine and Wellbeing, The University of Queensland |
| Project: | A combined program of education and exercise versus general advice for ankle osteoarthritis: A feasibility randomised controlled trial |
Recipients: Professor Bill Vicenzino, Associate Professor Michelle Smith, Dr Natalie Collins, Professor David Hunter, Dr Nathalia Costa
What question did the grant set out to answer?
Ankle osteoarthritis (OA) is a condition that is characterised by high levels of pain and disability[1]. It can affect individuals in all aspects of their lives, including work, recreation and socialisation [2]. Despite the serious impacts of ankle OA on people’s lives, there is limited research to how best to manage this condition. There are clinical practice guidelines to assist health professionals manage hip and knee OA, but nothing similar for ankle OA [3-6]. Generally, international guidelines on hip and knee OA management recommend education about the condition and how to manage it, weight loss (for people who are overweight or obese) and exercise. This research project applied these recommendations to ankle OA.
The main aim of this study was to determine the feasibility of running a full-scale randomised controlled trial investigating a combined education and exercise program compared to a general advice program for people with ankle OA. Secondarily, we aimed to collect data to inform sample size calculations and understand the perspectives of people with ankle OA on their participation in the interventions and project.
What did we do to answer this question?
We recruited 30 individuals with ankle OA from the community. To be eligible to participate in this study, individuals had to be: over 35 years of age, have ankle joint pain on most days for at least 3 months, rate their worst ankle pain in the past week as at least 3/10 on an 11-point numerical rating scale (NRS), have at least grade 2 (Kellgren-Lawrence) radiographic ankle OA on x-ray [7], and be able to understand English and commit to the allocated treatment. People with health problems or pain elsewhere that was more concerning than their ankle OA, previous ankle arthrodesis or joint replacement surgery, or other medical problems were not eligible to participate.
Participants in this study were randomised to receive either physiotherapist-delivered education and exercise, or physiotherapist-delivered general advice. The combined education and exercise program consisted of two group education sessions and 12 group exercise sessions over a 7-week period. The education sessions discussed what OA is, diagnosis and symptoms of ankle OA, international guidelines for the management of OA and evidence-based management of ankle OA. The exercise sessions included an aerobic exercise warm up on a stationary bike, exercises for foot/ankle muscle strength (e.g., resistance band and body weight exercises), compound lower limb exercises (e.g., step ups, squats, bridges), postural control/balance training (i.e., standing on compliant surfaces) and gentle stretching. Exercise resistance and difficulty was customised to the individual based on their ability. The general advice program consisted of a single information session about the importance of keeping active and tips on managing symptoms, based on online resources from Musculoskeletal Australia (Understanding Osteoarthritis) [8] and Arthritis Australia (Information Sheet on Physical Activity) [9].
We collected a range of outcome measures in the study. The primary outcome measure related to the feasibility of conducting a full-scale randomised controlled trial investigating a combined education and exercise program compared to a general advice program for people with ankle OA. We pre- emptively determined criteria to ascertain the feasibility of conducting a full-scale trial using the protocol from this grant. These feasibility outcomes were: a) Consent rate (percentage of consented participants from eligible individuals); b) Participant adherence with their allocated intervention (percentage of sessions attended); c) Physiotherapist fidelity delivering the intervention, evaluated by observation by a researcher and calculated as a percentage [10]; and d) Completion rate (percentage) of participants who completed the severity of ankle pain and Global Rating of Change outcome measures at 3 months. The feasibility criteria are outlined in Table 1. We also recorded number of responses to study advertisements, proportion of eligible individuals from interested individuals, recruitment rate per month, adverse events and proportion of completed outcome measures at 8 weeks. Participant perspectives on the credibility (how believable/logical the treatment is) and expectancy (expectations from treatment) of the intervention were evaluated at baseline, 2 weeks and 8 weeks after commencing the intervention [32].
Table 1. Feasibility criteria for a future randomised controlled trial
| Proceed | Proceed with caution | Do not proceed | |
| Consent rate | >50% | 30-50% | <30% |
| Participant adherence | >60% | 30-60% | <30% |
| Intervention fidelity | >60% | 30-60% | <30% |
| Completion of key PROMs at 3 months | >70% | 50-70% | <50% |
PROMs=participant-reported outcome measures
The secondary outcome measures in this study were used to calculate variability in measures. They included participant-reported outcomes measures collected at baseline, 8 weeks and 3 months after commencing the intervention, and physical outcomes collected at baseline and 8 weeks post- intervention commencement. The secondary outcome measures are described in Table 2.
Table 2. Secondary outcome measures.
| Outcome measure | Description |
| Global rating of change (GROC) | Perceived overall change in their ankle condition rated on a 7-point Likert scale [11]. |
| Ankle pain severity | Worst and average pain in the last week evaluated on an 11-point
numerical rating scale [11]. |
| Ankle stiffness severity | Worst and average stiffness in the last week evaluated on an 11-point numerical rating scale [11]. |
| Patient Acceptable
Symptom State (PASS) |
Participant’s opinion whether or not their current level of pain and
function is satisfactory (Yes/No) [12]. |
| Foot and Ankle Ability Measures (FAAM) | Self-report function during activities of daily living (ADL subscale) and sport (Sport subscale) [13]. |
| Foot and Ankle
Outcome Score (FAOS) |
Self-report measure of ankle pain, symptoms, function and quality of
life [14]. |
| Health-related quality of life (EQ-5D) | Health-related quality of life in relation to mobility, self-care, usual activity, pain/discomfort and anxiety/depression [15]. |
| Satisfaction | Satisfaction with treatment rated on a 5-point Likert scale. |
| 40-m walk test | Walking speed over a 40m distance (4 x 10m lengths) [16]. |
| Stairs descent test | Time taken to descend a flight of 10 stairs [1]. |
| Heel raise capacity test | Number of maximal height heel raises until fatigue [17]. |
| Ankle range of motion | The knee to wall test to measure ankle dorsiflexion range of motion during a weight-bearing lunge [18]. |
We conducted semi-structured interviews with participants at the end of the study to understand their perspectives on the intervention they were allocated to and their participation in the study.
What did we discover during the grant?
A total of 152 individuals responded to study advertisements, of which 31 (20%) were eligible to participate in the study. We recruited 30 participants over 8 months (~4 participants/month): 20 women (67%), median (inter-quartile range) age of 67 (59, 76) years, and body mass index of 29.9 (27.1, 35.7) kg/m2. 15 participants were randomised to the combined education plus exercise program, and 15 participants were randomised to the general advice program. There were no adverse events during the study. Over the course of the study, four participants (13%) withdrew from the study due to not having enough time (n=2) or managing other health issues (n=2).
Primary outcome data for the pre-determined feasibility criteria are shown in Table 3. Based on these feasibility criteria, we have determined it is feasible to run a full-scale randomised controlled trial investigating a combined education and exercise program compared to a general advice program for people with ankle OA.
Table 3. Data for feasibility criteria for a future randomised controlled trial
| % obtained | Indication | |
| Consent rate | 97% | Proceed |
| Participant adherence | 71% | Proceed |
| Intervention fidelity | 94% | Proceed |
| Completion of key PROMs at 3 months | 77% | Proceed |
PROMs=participant-reported outcome measures
Data on participants’ perspectives on the credibility and expectancy of their allocated intervention is shown in Table 4 for each group.
Table 4. Credibility and expectancy data (data presented as median (inter-quartile range) unless otherwise specified)
| Outcome | Education + exercise program | General advice program | ||||
| Baseline (n=12) | 2 weeks (n=12) | 8 weeks (n=11) | Baseline (n=14) | 2 weeks (n=13) | 8 weeks n=12) | |
| Credibility | ||||||
| How logical the treatment seems, /9 | 8 (5, 9) | 9 (9, 9) | 9 (9, 9) | 6 (5, 7) | 5 (4, 8) | 5 (2, 6) |
| How successful they think
it will be, /9 |
5 (5, 7) | 7 (7, 7) | 7 (6, 9) | 6 (5, 8) | 4 (3, 5) | 4 (2, 5) |
| Confidence recommending to a friend, /9 | 5 (5, 7) | 8 (7, 8) | 9 (8, 9) | 5 (5, 6) | 4 (3, 5) | 4 (1, 5) |
| How much improvement in symptoms they think
will occur, /100 |
45
(28, 50) |
60
(38, 70) |
50
(20, 75) |
40
(20, 50) |
20
(10, 20) |
5
(0, 25) |
| Expectancy | ||||||
| How much they feel the treatment will help, /9 | 5 (5, 7) | 7 (5, 8) | 6 (6, 8) | 5 (5, 6) | 4 (2, 5) | 3 (1, 5) |
| How much improvement in symptoms they feel will
occur, /100 |
45
(28, 53) |
55
(38, 70) |
50
(30, 70) |
40
(20, 50) |
20
(10, 20) |
5
(0, 23) |
A higher number indicates greater perceived credibility and higher expectations.
Data for the participant reported and physical outcome measures collected throughout the study for the combined exercise and education group and general advice group are shown in Table 5.
Table 5. Secondary outcome measures as median (inter-quartile range) unless otherwise specified)
| Outcome | Education + exercise program | General advice program | ||
| Baseline (n=12) | 8-weeks (n=11) | Baseline (n=14) | 8-weeks (n=12) | |
| GROC, % (n) | ||||
| Much better | NA | 18.2 (2) | NA | 8.3 (1) |
| Better | NA | 27.3 (3) | NA | 8.3 (1) |
| Slightly better | NA | 45.5 (5) | NA | 16.7 (2) |
| Same | NA | 9.1 (1) | NA | 50.0 (6) |
| Slightly worse | NA | 0 (0) | NA | 8.3 (1) |
| Worse | NA | 0 (0) | NA | 8.3 (1) |
| Much worse | NA | 0 (0) | NA | 0 (0) |
| Worst pain, /10 | 5.5 (4.8, 6.3) | 3.0 (2.0, 4.5) | 7.0 (6.3, 8.0) | 5.5 (2.0, 8.0) |
| Average pain, /10 | 2.5 (2.0, 5.3) | 2.0 (1.0, 3.0) | 4.0 (3.0, 5.0) | 4.0 (2.0, 5.0) |
| Worst stiffness, /10 | 5.0 (2.0, 6.5) | 3.0 (2.0, 4.0) | 7.0 (5.3, 7.0) | 6.0 (3.0, 7.3) |
| Average stiffness, /10 | 2.5 (1.8, 4.3) | 2.0 (1.0, 3.0) | 5.0 (3.0, 6.0) | 5.0 (2.8, 6.0) |
| PASS, % (n) yes | 8.3 (1) | 63.6 (7) | 21.4 (3) | 50.0 (6) |
| FAAM-ADL, % | 80.4 (60.7,
86.0) |
85.7
(72.1, 94.7) |
64.9
(61.9, 72.0) |
69.6
(62.5, 81.3) |
| FAAM-Sport, % | 57.8
(45.8, 68.8) |
65.6
(42.2, 78.1) |
43.8
(29.2, 52.3) |
50.0
(31.3, 73.4) |
| FAOS symptoms, /100 | 53.6
(46.4, 65.2) |
62.5
(56.3, 62.5) |
50.0
(46.4, 53.6) |
57.8
(49.2,68.8) |
| FAOS pain, /100 | 72.2
(61.1, 77.8) |
69.4
(66.7, 84.7) |
55.6
(52.8, 72.2) |
73.6
(44.4, 81.3) |
| FAOS ADL function,
/100 |
84.6
(74.3, 94.9) |
76.5
(70.6, 91.2) |
73.5
(70.6, 83, 8) |
77.2
(66.9, 88.6) |
| FAOS sport/
recreation, /100 |
67.5
(55.0, 83.8) |
75.0
(42.5, 87.5) |
60.0
(50.0, 75.0) |
62.5
(45.0, 73.8) |
| FOAS QoL, /100 | 46.9
(35.9, 51.6) |
50.0
(43.8, 62.5) |
37.5
(31.3, 50.0) |
50.0
(31.3, 64.1) |
| QoL EQ-5D UK Index,
/1 |
0.88
(0.81, 0.94) |
0.92
(0.82, 0.95) |
0.81
(0.75, 0.83) |
0.85
(0.77, 0.94) |
| QoL EQ-5D VAS, /100 | 82.5 (70.0,
91.0) |
85.0
(77.0, 90.0) |
75.0
(66.0, 90.0) |
83.5
(71.5, 86.3) |
| Satisfaction, % (n) | ||||
| Very | NA | 72.7 (8) | NA | 8.3 (1) |
| Somewhat satisfied | NA | 27.3 (3) | NA | 25 (3) |
| Neither satisfied or
dissatisfied |
NA | 0 (0) | NA | 58.3 (7) |
| Dissatisfied | NA | 0 (0) | NA | (8.3) (1) |
| Very dissatisfied | NA | 0 (0) | NA | 0 (0) |
| 40-m walk, s b | 25.0 (21.5,
28.8) |
24.9 (20.4,
26.7) |
26.4 (22.8, 30.4) | 26.3 (21.8, 31.3) |
| Stairs descent, s | 4.9 (3.8, 5.2) | 4.2 (3.3, 5.5) | 4.7 (3.7, 6.2) | 5.1 (3.9, 5.5) |
| Heel raise capacity, # | 11 (7, 19.5) | 15 (7, 20) | 11 (7, 13.5) | 8.5 (3.5, 12.5) |
| Dorsiflexion ROM, cm | 7.2 (5.6, 13.0) | 6.5 (6.0, 11.9) | 10.5 (9.6, 12.5) | 10.5 (4.8, 12.4) |
GROC=global rating of change, PASS=patient acceptable symptom scale, FAAM=foot and ankle ability measures, ADL=activities of daily living, FAOS=foot and ankle outcome score, QoL=quality of life, VAS=visual analog scale ROM=range of motion, NA=not applicable
Have the findings of the research already benefitted people with musculoskeletal disease?
We have published the protocol for this study in which we have fully described the education and exercise intervention. This provides information that health professionals can use when managing patients with ankle OA.
As this was a feasibility study, it did not compare the effectiveness of the two interventions studied (combined education and exercise program vs a general advice program). Its findings indicate conducting a full scale clinical trial to test the effectiveness of the intervention is feasible – a likely benefit for people with ankle OA.
What are the next steps for this research?
Based on this grant demonstrating that it is feasible to run a full-scale trial investigating a combined education plus exercise program compared to a general advice program for people with ankle OA, we will begin planning for a full-scale randomised controlled trial. We will evaluate perspectives of study participants on their participation in the interventions and project – from semi-structured interview data – to inform if adjustments to the protocol are needed before designing and undertaking a full- scale trial. We will apply for funding to undertake a randomised controlled trial based on findings from this feasibility trail..
What is the plan for dissemination of findings?
We have published the protocol for this research study in the Journal of Foot and Ankle Research.
Smith MD, Vuvan V, Collins NJ, Hunter DJ, Costa N, Smith MMF, et al. Protocol for a randomised feasibility trial comparing a combined program of education and exercise versus general advice for ankle osteoarthritis. J Foot Ankle Res. 2023;16:72. doi: 10.1186/s13047-023-00669-1.
We have also presented the protocol and rationale for this study at the following conferences:
Smith MD, Smith MM. Ankle Osteoarthritis: Improving skills in managing this commonly forgotten condition. Sports Medicine Australia, October 2023.
Smith MD. Managing post-traumatic ankle osteoarthritis. Queensland Musculoskeletal Outpatients Physiotherapy Network Conference, November 2023.
We plan to publish the feasibility study in an international peer-review journal and disseminate findings on social media and through our professional networks.
We would like to thank Arthritis Australia for making this research possible and supporting the progression of research to establish evidence-management for ankle OA.
References
- Al-Mahrouqi MM, Vicenzino B, MacDonald DA, Smith Disability, Physical Impairments, and Poor Quality of Life, Rather Than Radiographic Changes, Are Related to Symptoms in Individuals With Ankle Osteoarthritis: A Cross-sectional Laboratory Study. J Orthop Sports Phys Ther. 2020;50(12):711-22.
- Alanazi SA, Vicenzino B, Maclachlan LR, Smith MD. “It’s like a nail being driven in the ankle”: A qualitative study of individuals’ lived experiences to inform a core domain set for ankle osteoarthritis. Musculoskelet Sci 2023;66:102813.
- 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee (vol 72, pg 149, 2020). Arthritis & Rheumatology. 2021;73(5):799.
- National Institute for Health and Care Excellence. Osteoarthritis in over 16s: diagnosis and management NICE guideline [NG226]. UK;
- The Royal Australian College of General Practitioners. Guideline for the management of knee and hip osteoarthritis (2nd edition). East Melbourne, Vic: RACGP;
- Zhang W, Nuki G, Moskowitz RW, Abramson S, Altman RD, Arden NK, et OARSI recommendations for the management of hip and knee osteoarthritis: part III: Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis Cartilage. 2010;18(4):476-99.
- Kraus VB, Kilfoil TM, Hash TW, 2nd, McDaniel G, Renner JB, Carrino JA, et Atlas of radiographic features of osteoarthritis of the ankle and hindfoot. Osteoarthritis Cartilage. 2015;23(12):2059-85.
- Musculoskeletal Understanding Osteoarthritis. Elsternwick, VIC: Musculoskeletal Australia; 2022.
- Arthritis Arthritis Australia info sheet Physical activity. Glebe, NSW: Arthritis Australia; 2007.
- Davis AM, Kennedy D, Wong R, Robarts S, Skou ST, McGlasson R, et al. Cross-cultural adaptation and implementation of Good Life with osteoarthritis in Denmark (GLA:D (TM)): group education and exercise for hip and knee osteoarthritis is feasible in Osteoarthritis Cartilage. 2018;26(2):211-9.
- ten Klooster PM, Drossaers-Bakker KW, Taal E, van de Laar Patient-perceived satisfactory improvement (PPSI): interpreting meaningful change in pain from the patient’s perspective. Pain. 2006;121(1-2):151-7.
- Kvien TK, Heiberg T, Hagen KB. Minimal clinically important improvement/difference (MCII/MCID) and patient acceptable symptom state (PASS): what do these concepts mean? Ann Rheum Dis. 2007;66 Suppl 3:iii40-1.
- Martin RL, Irrgang JJ, Burdett RG, Conti SF, Van Swearingen JM. Evidence of validity for the Foot and Ankle Ability Measure (FAAM). Foot Ankle 2005;26(11):968-83.
- Roos EM, Brandsson S, Karlsson J. Validation of the foot and ankle outcome score for ankle ligament Foot & ankle international / American Orthopaedic Foot and Ankle Society [and] Swiss Foot and Ankle Society. 2001;22(10):788-94.
- Rabin R, de Charro EQ-5D: a measure of health status from the EuroQol Group. Ann Med. 2001;33(5):337- 43.
- Wright AA, Cook CE, Baxter GD, Dockerty JD, Abbott JH. A Comparison of 3 Methodological Approaches to Defining Major Clinically Important Improvement of 4 Performance Measures in Patients With Hip J Orthop Sport Phys. 2011;41(5):319-27.
- Hebert-Losier K, Wessman C, Alricsson M, Svantesson U. Updated reliability and normative values for the standing heel-rise test in healthy Physiotherapy. 2017;103(4):446-52.
- Smith MD, Lee D, Russell T, Matthews M, MacDonald D, Vicenzino B. How Much Does the Talocrural Joint Contribute to Ankle Dorsiflexion Range of Motion During the Weight-Bearing Lunge Test? A Cross-sectional Radiographic Validity J Orthop Sports Phys Ther. 2019;49(12):934-41.
Clinical Fellowship in Inflammatory Myositis and Interstitial Lung Disease and Data Science
| Funded by: | ARA Research Fund |
| Recipient: | Dr Aravinthan Loganathan |
| Intended Department | Rheumatology Department, Royal National Hospital of Rheumatic Diseases, Bath & Department of Life Sciences, University of Bath |
| Project: | Clinical Fellowship in Inflammatory Myositis and Interstitial Lung Disease and Data Science |
Overview
I completed an overseas research fellowship at the Royal National Hospital of Rheumatic Diseases Bath, United Kingdom and Department of Life Sciences, University of Bath, with the support of the Ken Muirden Fellowship and Arthritis Australia.
The Ken Muirden fellowship provided me with the opportunity to delve into the field of inflammatory myositis, an area experiencing dynamic growth within rheumatology due to the identification of new myositis antibodies and a deeper understanding of the disease’s multi-systemic nature, encompassing cutaneous, pulmonary, and cardiac manifestations. My focus was inflammatory myositis and interstitial lung disease, significantly contributing to morbidity and mortality.
The collaboration with the University of Bath and the Royal National Hospital of Rheumatic Diseases provided a conducive environment for advancing my research and clinical capabilities. Throughout my fellowship, I engaged in research and clinical endeavours, improving my expertise and understanding of these complex conditions. Moreover, this fellowship allowed me to work with international experts and researchers in inflammatory myositis. Also, it allowed me to develop international research links, which I aim to utilise in future research projects.
Additionally, this fellowship presented the unique and invaluable opportunity to gain experience in laboratory work in autoantibody testing, gaining hands-on experience in the interpretation and basic science of various immunological tests utilised in rheumatology, specifically those about myositis. This exposure significantly enhanced my clinical reasoning and judgment, offering a more nuanced understanding of the strengths and limitations inherent in immunological antibody testing.
Detecting different myositis-specific and myositis-associated antibodies is pivotal to diagnosing diverse idiopathic inflammatory myopathies. Incorrect interpretation of these antibodies can lead to unnecessary investigations, follow-up and treatment. I aim to use the experiences this fellowship has provided me in my future practice.
Research Accomplishments/Projects:
During my fellowship, I was involved in different research projects. I presented these research projects at conferences during my time in Bath at various meetings. My research projects, listed below, focussed on autoantibody detection in myositis and clinical management and evaluation of interstitial lung disease in myositis. These projects involved working with different international groups, allowing me to understand the differences in the clinical presentations in evaluating and treating myositis in other population groups.
- Global Conference on Myositis Prague 2022 (DOI: 10.55563/clinexprheumatol/zka1xq)
- Abstract/Poster: Detection of Anti-Zo and Other rare anti-synthetase syndrome autoantibodies on research line blot immunoassays
- This project evaluated the accuracy of a new myositis research line blot immunoassay in detecting antibodies to anti-synthetase autoantibodies anti-Zo, Ha and Ks.
- We used newly developed line blot immunoassays to detect these antibodies and successfully demonstrated that anti-Zo was detected in all cases; however, we could not detect cases of anti-KS and anti-Ha.
- Abstract/Poster: Detection of myositis autoantibodies in patients labelled with idiopathic interstitial lung disease
- Using line blot on sera samples of patients with idiopathic interstitial lung disease in which CTD had not been diagnosed. We demonstrated on IP and confirmed with the more commonly used line blot tests that antibodies associated with connective tissue disease and myositis were readily accessible, highlighting the importance of developing a testing strategy for cases of idiopathic ILD.
- British Society Rheumatology Annual Meeting 2023: (DOI: 10.1093/rheumatology/kead104.195)
- Abstract/Poster: Real-world audit of patients with idiopathic inflammatory myositis-related interstitial lung disease in a European population
- The RNHRD operates specialist clinics for scleroderma and lupus, but IIM patients are currently seen in a general CTD clinic. As part of work to establish a dedicated myositis clinic, we collated data on 71 patients under active follow-up with myositis spectrum disease deemed appropriate for follow-up within a specialist clinic.
- We reviewed patients diagnosed with IIM-ILD and reviewed the serological immune profile, screening practices for pulmonary involvement, findings on CT/ pulmonary function tests and pharmacological management.
- Our review demonstrates that 65% of patients with adult IIM were screened for ILD. NSIP and OP were the most common radiological appearances. Half of our cohort required aggressive immunosuppression, cyclophosphamide being the most prescribed agent and mycophenolate being preferred for maintenance therapy. No patients were prescribed tacrolimus or ciclosporin. Despite treatment, 52% of patients had evidence of progressive disease on serial PFTs
- EULAR 2023 (DOI:10.1136/annrheumdis-2023-eular.2218)
- Abstract (Publication Only): Clinical Phenotype and Antibody Correlations using Immunoprecipitation in Indian patients with idiopathic inflammatory myopathies from the Myocite cohort.
- We reviewed the clinic-serological profile of 148 patients from a well-characterised MyoCite cohort of juvenile and adult patients with IIM from Lucknow, India, to determine significant clinical characteristics associated with different myositis-specific antibodies.
- Conclusion: Autoantibodies are present in nearly two-thirds of Indian patients with IIM, although the prevalence of individual MSA differs. The frequency of ILD is closer to those previously reported in European cohorts than in East Asian populations.
- Most MSAs exhibited association with prominent cutaneous manifestations, including mechanics hands, heliotrope rash, Gottron’s sign, V-sign, hyperpigmentation and erythematous changes.
- These differences in antibody profile and clinical characteristics from the MyoCite cohort compared to those of different geographical and ethnic populations previously reported suggest the potential influence of genetics and/or environmental factors.
- Journal Article: Frontiers in Immunology (DOI: 3389/fimmu.2022.975939)
- Title: The use of ELISA is comparable to immunoprecipitation in the detection of selected myositis-specific autoantibodies in a European population
- We evaluated the use of ELISA in comparison to IP for different myositis-specific antibodies (Mi2, MDA5, Jo1, EJ, KS, PL7, PL12)
- Our paper demonstrated a very good agreement between ELISA and IP, suggesting that ELISA can detect these antibodies and aid diagnosis and is comparable to the reference standard immunoprecipitation.
- Abstract (Publication Only): Clinical Phenotype and Antibody Correlations using Immunoprecipitation in Indian patients with idiopathic inflammatory myopathies from the Myocite cohort.
- Abstract/Poster: Real-world audit of patients with idiopathic inflammatory myositis-related interstitial lung disease in a European population
- Abstract/Poster: Detection of Anti-Zo and Other rare anti-synthetase syndrome autoantibodies on research line blot immunoassays
Research Accomplishments/Projects to be completed:
Regrettably, I could not conclude all the projects initiated during my fellowship. Some of these projects are presently in the review process with various collaborators and are slated for submission to pertinent journals. Here is a concise summary of these ongoing projects:
CLASS Project (Junior Researcher):
- “Agreement between local and central anti-synthetase antibodies detection: results from the CLASS project biobank.”
- Evaluation of patients with Interstitial Lung Disease (ILD) in Anti-Synthetase syndrome within the CLASS database – currently undergoing evaluation/research.
- Evaluation of muscle biopsy findings in patients with the anti-synthetase syndrome – currently undergoing evaluation/research.
MyoCite Research Group:
- Assessing the Sensitivity and Specificity of Myositis-Specific and Associated Autoantibodies: A Sub-study from the MyoCite cohort
Royal National Hospital of Rheumatic Diseases:
- ILD and use of Patient Reported Outcome Measures – Engaged in a systematic review of diverse scoring systems employed to assess symptoms in evaluating ILD – work in progress and to be completed.
Clinical Skills & Data Science:
This fellowship gave me a unique opportunity to enhance my clinical proficiency and broaden my myositis, general rheumatology, and data science skillset. Additionally, it afforded me valuable experience in establishing a new clinical service. In greater detail, these experiences encompassed:
- Musculoskeletal Ultrasound:
- Proficient in conducting ultrasound scans, with expertise in detecting subclinical synovitis in the hands and feet
- Setting up follow-up clinics and clinical database:
- Helped establish a patient-initiated follow-up clinic, extending outpatient follow-up appointments for rheumatology in order to address the delayed waiting list times post COVID-19. This pathway provided a safe avenue for clinicians to delay the follow-up of stable patients, allowing patients who were overdue or needed earlier to review to be seen more quickly. The RNHRD was the first centre to enrol 5% of its outpatient follow-up patients in a PIFU pathway in the NHS.
- Experience in collaborating with key stakeholders such as medical staff, hospital administration, coding/statistical staff, nursing & outpatient staff, and General Practitioners.
- Creation of follow-up pathways, including arrangements for patients requiring expedited follow-up and providing training and education to staff members who were likely to be the point of contact for patients needing earlier follow-up
- Implemented a comprehensive database for data collection, identifying suitable patients specifically for the myositis clinic at the RNHRD.
- Data Science
- Experience in performing statistical analysis in evaluating the performance of different antibodies as well as performing calculations to find significant associations for different variables associated with different antibodies.
- Muscle Biopsy:
- Experience performing open muscle biopsies on lower limbs for diagnostic purposes.
- Clinical Experiences:
- Participated in combined rheumatology/respiratory clinic meetings to enhance understanding and treatment of CTD-ILD, focusing on myositis-related ILD.
- Worked in rheumatology connective tissue disease at the RNHRD, Bath, gaining valuable experience treating myositis.
- Laboratory Experience:
- Developed proficiency in performing various immunological tests, including ELISA and line blot, and gained observational experience in immunoprecipitation procedures.
- Deepened understanding of the scientific principles underlying these tests and acquired insights into the potential pitfalls in their clinical application
Summary:
I am grateful for the support extended to me by Arthritis Australia and the Ken Muirden fellowship during my overseas fellowship. This invaluable experience has laid a robust foundation for my research and clinical career. I am committed to utilising the skills gained to enhance the care provided to individuals with inflammatory myositis and other rheumatological conditions.
A Qualitative Study to define Independence in the context of Rheumatoid Arthritis remission from the Patient Perspective
| Funded by: | ARA Research Fund |
| Recipient: | Dr Kim Griggs |
| Intended Department | Rheumatology Unit, Royal Adelaide Hospital |
| Project: | A Qualitative Study to define Independence in the context of Rheumatoid Arthritis remission from the Patient Perspective |
Overview
The research team would like to thank Arthritis Australia for their support with this project. This grant has enabled us to complete focus groups/interview to identify the full range of issues from a patient’s perspective that define independence in the context of remission in rheumatoid arthritis. This is the first qualitative study to focus on independence in the context of RA remission from the patient perspective. Going forward, patient and professional consensus on the most important issues raised in these focus groups will be sought in order to develop a measure for independence in the context of RA remission.
Background
Rheumatoid arthritis (RA) is a systemic, inflammatory condition causing synovitis and pain in multiple joints, which can result in permanent disability with accompanying emotional, social, financial, and societal burden. Remission (a state of ‘disease activity as good as gone’) is the desired outcome in RA treatment. However, the current American College of Rheumatology/European Alliance of Associations for Rheumatology (ACR/EULAR) remission criteria have previously been criticised for not adequately incorporating the patient perspective.
Qualitative focus groups were conducted to identify what remission meant to people with RA, which identified a list of 26 domains that were then ranked in order of importance in a survey study with RA patients from six countries. Domains most frequently included in the top three by participants were: pain, fatigue, and independence.
RA patients with low disease activity or in patient-perceived remission completed validated surveys measure pain and fatigue, and a numerical rating scale was created by the researchers to measure independence. The validated pain and fatigue numerical rating scales worked well to assess RA remission. However, the new independence numerical rating scale worked well with stable disease activity but did not perform as well in predicting change in disease activity.
These results indicated that the domain of independence is worthy of further exploration. To inform the development of a validated patient reported outcome measure we first need to understand how patients are using and defining the concept of Independence in the context of RA remission.
What question did the grant set out to answer? What problems did you try to solve, or gaps in the knowledge did you try to fill? Why is this important?
A qualitative systematic literature review found mentions of independence could be organised into themes of: a return to state before arthritis; being physically and functionally able; a sense of freedom without needing to rely on others; and having control over the organisation of one’s life. However, none of these studies specifically asked patients to focus on independence and could therefore be missing key information to define independence. Thus, we aimed to understand independence in the context of rheumatoid arthritis remission from the patient perspective through further qualitative research.
The main objectives of this grant were to:
- Establish a patient-centred definition of ‘Independence’ in the context of rheumatoid arthritis remission.
- Clarify whether ‘Independence’ is the correct term for this concept.
- Explore which aspects of their condition patients consider when completing the patient global assessment when in low disease activity.
Focus groups and interview:
Nineteen RA patients participated in focus groups, or an interview designed to promote discussion around themes identified by the systematic literature review. New concepts raised by participants were explored in subsequent focus groups.
What were the main findings of the research?
Five distinct but interconnected themes were identified reflecting how patients are using and defining the concept of Independence in the context of RA remission: physically and functionally able, participation beyond function, cognitive independence, assistance is complicated, having or taking control.
“I can get myself dressed”: Physically and functionally able
To many participants independence included being physically and functionally able to do the things they wanted to across activities of daily living, meeting their responsibilities, and engaging in valued life activities. These activities could be grouped into themes.
- Activities of daily living: this included being able to do personal care tasks for themselves. For some participants the independence of being able to do these daily tasks was more important than levels of pain to their experience of remission.
- Meeting responsibilities: this meant being able to run their own errands without help, being able to continue working with the tasks associated with that and being able to drive to fulfil responsibilities without help from others.
- Valued activities: being physically and functionally able to participate in valued activities that go beyond regular day to day activities. These activities included quality time with family and friends and the ability to participate in social activities and the ability take part in active hobbies and pursuits through exercise or fun activities that may be physically demanding. Participants highlighted the importance of measuring how many things have been missed due to RA rather than simply measuring what patients have been able to do. Participants emphasised the impact of independence on their mental health; poor mental health could lead to feeling stuck and unable to participate, whilst lack of functional ability to participate could also negatively affect mental health.
“Something more substantial”: Participation beyond function
Whilst function was discussed as an important component to being independent, many participants emphasised that independence is more than being able to carry out physical tasks.
- Beyond being present: independence in the context of remission meant going beyond simply being able to attend events or carry out tasks. Participants reported needing to feel productive and explained the importance of not having to struggle to do the things they wanted to. To some participants being independent meant being able to do all the components of a task (for example in the workplace) rather than needing help with certain aspects.
- Confidence to make future plans: the ability to make future plans without worrying whether they would be well enough. Being able to be relied upon by others was an important aspect of independence. Cancelling plans was cited as a clear sign that they were not in remission.
- Not having to pace or be selective: Pacing and planning is an important aspect of RA self-management, and to some participants independence meant being free of having to consider this. Participants wanted to do the things they wanted to, and have something left afterwards without having to consider planning recovery time. Some participants explained that being able to do things without having to pace was another important distinction between being stubbornly independent despite disease activity and being independent in the context of RA remission.
- Being ‘normal’, but what is ‘normal’? Many participants reported that independence meant being back to ‘normal’ with some suggesting this as the goal on a scale for independence. However, there was not agreement between participants as to what ‘normal’ meant. Opinions ranged from being able to do everything they could do before having RA , being as independent or being able to keep up with peers or fitting in with societal expectations of participation in daily activities. However, some found it hard to distinguish what independence may have been lost naturally due to aging and what ‘normal’ meant in a healthy population where fitness levels can vary widely.
“It’s a mind battle”: Cognitive independence
Most participants discussed defining independence as being not just physically but mentally independent of RA. This meant being independent of having to constantly think or plan ahead, free of the worries that come with RA, not having the mental burden of managing the disease, and the most desirable form of cognitive independence was being able to forget they have RA.
- Not having to think ahead: Participants explained the mental exhaustion they experienced through having to always think ahead to accommodate RA disease activity. The ability to be spontaneous without needing back up plans in place was an important component of independence.
- Not thinking about RA management: Participants discussed the mental load of having to plan their life around hospital appointments and medication. To many participants defining independence meant being free of all the aspects of disease management that reminds them of having RA. Whilst some participants were satisfied that they could be independent whilst still on medication, for others true independence meant also being free of having to think about drug regimes or worry about the related side effects.
- Forgetting about RA: For many participants the most desirable form of independence was being able to forget about RA completely, which included being well enough to not be reminded of their RA through other people’s reactions.
“Somebody’s going to have to define assistance”: Assistance is complicated
A key concern across participants for including assistance in a definition or measure of independence was that assistance means different things to different people. The scale of assistance was seen as a broad spectrum, making it difficult to understand what a generic measure of assistance might be referring to. Participants felt it was important to note that there is a difference between needing assistance and receiving it as another person happens to be with you, and highlighted that some assistance may be no more than a healthy population might need.
For consideration in creating a measure of independence for remission that includes assistance some participants noted that assistance may have a complicated relationship with independence. First, patients may still consider themselves independent when receiving assistance depending on their perception. Second, lack of assistance may not be an accurate indication of remission as patients may have adjusted their routines or adapted their lives to work around the need for assistance.
- Everyone needs help sometimes: Some participants embraced assistance as part of life, not something they needed specifically due to RA. They noted that everyone needs assistance to some extent, and found it difficult to clarify where assistance was needed specifically due to RA. Tools and devices were seen by some as an aid to independence, rather than challenging their self-perception of being independent. Similarly, medication was seen by many participants as a way to aid their independence rather than impeding it.
- Not reliant on anyone or anything: For some participants independence meant not being reliant on other people, tools and devices or medications to help them go about their daily lives. Some took this to an extreme with independence for them being free of any help at all. Participants reported that not needing help from other people was an important element of independence.
“A degree of personal autonomy”: Having or taking control
To many participants independence meant having control of their lives and the decisions they make. This meant having control of their life choices and taking back control through pushing for independence regardless of RA symptoms.
- Choices not dictated by RA: An important wider element of independence to participants was being able to make their own decisions independent of RA. This ranged from everyday decisions such as which clothes they chose, to being able to plan for a family without needing to consider their medication or discuss with their clinical team. Participants talked about the importance of having autonomy over their lives, which seemed to be the gold standard for independence.
- Independence regardless of RA: An important consideration for a measure of independence in the context of RA remission is that some participants reported finding ways to be independent despite the challenges RA brings. This sometimes meant pushing themselves to their limits and could mean independence being prioritised over pain or joint damage. Some participants highlighted that barriers to taking control of their lives or doing things without other people may be things that healthy populations may also struggle with. It is possible that to an extent independence is a mindset.
Underpinning theme: Stages of independence
Underpinning these findings is the idea that independence meant slightly different things to everyone. Participants suggested “there are stages of independence”, each of which may be interpreted in their own right to mean independence, and there may be other factors beyond disease activity that hold patients in each of these stages, such as disability, personal circumstances, and intrinsic motivation. Patients can be ’practically independent’, where they may not have all the function to do everything in the way they used to or want to, but they have adapted and found work-arounds to still enable them to do things they want to. The next stages are ‘functionally independent’ (functionally able to take part in the things they want to); ‘socially independent’ (beyond presenteeism, and without the need to pace); ‘cognitively independent’ (not having to plan ahead, think or worry about RA) and ‘medically independent’ (free of medical management). Finally, full autonomy was seen as the gold standard for independence, being in control of all aspects of life without having to consider RA.
All of these distinct stages could be interpreted by patients to mean Independence, emphasizing the importance of creating a measure that captures Independence within the context of interest as simply asking patients if they are independent is too subjective. Depending on how participants interpreted independence, whilst some felt independent despite limitations of RA, others questioned whether true independence with RA was ever possible.
How might the findings inform further research?
This is the first qualitative study to focus on independence in the context of RA remission from the patient perspective. These novel data on defining and measuring independence in RA remission suggest this is a complicated concept that will require a more sophisticated measure than a single scale. It is likely a composite measure will need to be developed that goes beyond function and takes into account the multi-dimensional nature of independence. Next steps will be to seek patient and professional consensus on the most important issues raised in these focus groups to be taken forward to developing a measure for independence in the context of RA remission.
Have you disseminated or plan to disseminate the results of this research?
Two manuscripts related to this work are in preparation for submission to peer reviewed journals:
- Flurey CA, Jones B, Gazel U, Uzoka C, Rosser K, Khoo T, Scholte M, Hoogland W, Shea B, March L, Beaton D, Tugwell P, Proudman S. A qualitative study to define independence in the context of rheumatoid arthritis remission from the patient perspective.
- Gazel U, Khoo T, Jones B, Tugwell P, Proudman S, Flurey CA. Remission independence OMERACT SIG proceedings.
Validation and integration of Patient Reported Experience Measures in outpatient rheumatology care in Australia
| Funded by: | ARA Research Fund |
| Recipient: | Dr Madeleine Bryant |
| Intended Department | The Queen Elizabeth Hospital, Department of Rheumatology |
| Project: | Validation and integration of Patient Reported Experience Measures in outpatient rheumatology care in Australia |
Overview
The research team have been extremely thankful for the opportunity to undertake this project with the support of an Arthritis Australia Grant In Aid. We believe the outcomes of our project will have a tangible effect on improving the experience of care that our patients receive when attending rheumatology clinics across Australia. We have modified and validated a Patient Reported Experience Measure (PREM) with Australian patients, meaning this instrument may now be used to routinely capture experience-related data. Sitting alongside other outcome measure instruments, the modified PREM will enable providers to qualify factors relating to the process, content and impact of care, directly capturing the perspectives of consumers.
Phase 1
The objectives of Phase 1 of this project have been fully realised and published in rheumatology literature [1]. In this work, we sought to capture the current lived experience of patients and healthcare professionals receiving and providing care in rheumatology clinics, and to explore patient and health-care professional (HCP) views on what constitutes high quality, patient-centred care. This is a foundational component of establishing the role and remit of a purpose-designed PREM to routinely build experience-related data into quality improvement systems within rheumatology as a specialty in Australia.
We conducted a non-interventional, qualitative study comprising five semi-structured, facilitated focus groups. Participants included rheumatology outpatients (n=16) of two tertiary teaching hospitals (The Queen Elizabeth and Royal Adelaide Hospitals), and healthcare professionals (n=14) (rheumatologists, rheumatology trainees, physiotherapists, specialty nurse, pharmacist). Participants explored priorities when attending outpatient services, real experiences, and aspirations for improving future care. Focus groups were audio-recorded, professionally transcribed verbatim, and coded using inductive and deductive thematic analysis by the research team [2-4]. Conceptual links between individual perspectives were explored both within and between the patient and clinician groups. Potential key themes were identified as those repeatedly raised and explored in depth during the focus groups [4]. The study was approved by the Central Adelaide Local Health Network Human Ethics Committee (reference 13846). Written informed consent was obtained, including consent for audio recording and transcription.
Results: Seven key themes were identified. These included smooth flow of technical processes, care coordination, individualised care, information sharing, clinical excellence, patient empowerment and comprehensive care. Such themes were conceptually aligned with quality standards in Australia and worldwide [5-7]. Overall we identified that patient and HCP cohorts are concerned with the same issues, though prioritisation is different between groups. Several disparities were noteworthy; the foremost of which being a discord in appreciation of the impact on logistics on the overall care experience. To this end, the most highly prioritised themes for patients pertained to processes and technical aspects of care. Issues of waiting time and appointment inflexibility were demonstrated as universal patient concerns, and their negative impact on care was grossly evident in patient cohort data. HCPs focused on themes relating to non-technical aspects of service provision: another noteworthy difference in prioritisation and conceptualisation between cohorts. Information sharing, individualisation of care, patient advocacy and empowerment were frequently occurring and deeply explored themes in the HCP cohort. HCPs described many different manifestations of empowerment, such as the importance of facilitating patient selfadvocacy, acknowledging patient ownership of health information, and adjusting care to account for individual health literacy. This theme was explored in detail by HCPs and was given high priority in descriptions of high quality care, but was not as strongly evident in patient focus group data. Participants from both patient and HCP cohorts highlighted the importance of mutual respect and pursuit of therapeutic partnership.
Summarily, this study captured valuable insights into the current experience of outpatient rheumatology care from the perspective of patients and healthcare professionals. It informs a collective understanding of differing and shared priorities, positives of current care, and areas requiring improvement.
Citation: Bryant MJ, Munt R, Black RJ, Reynolds A, Hill CL. Joining forces to understand what matters most: qualitative insights into the patient experience of outpatient rheumatology care, Rheumatol Adv Practice, 2023, rkad068, https://doi.org/10.1093/rap/rkad068
This work has been distributed in the following forums:
- Oral presentation, Patient Reported Measures Research Symposium, Commission for Excellence and Innovation in Health (CEIH), Adelaide, 18 March 2022.
- Oral presentation, Australian Rheumatology Association Annual Scientific Meeting, Sydney, 20 May 2022. Awarded Best Clinical Free Paper.
- Oral presentation, Basil Hetzel Institute Research Expo, Adelaide, 21 Oct 2022. Awarded best oral presentation, best lay summary.
- Online report, The Limbic rheumatology. Available at https://thelimbic.com/rheumatology/what-do-rheumatology-patients-value-mostduring-clinic-visits/
Phase 2
Data collection and analysis for Phase 2 of this project have been realised, with a report now in the editing phase and anticipated to be submitted for publication by end 2023.
In this project, we undertook the necessary steps to evaluate the reliability and validity of an adapted version of the Commissioning for Quality in Rheumatoid Arthritis-PREM (CQRAPREM), for assessment of the patient care experience in an Australian mixed rheumatology patient cohort. Building on the findings of Phase 1 work, we identified that the priorities and concerns of Australian rheumatology patients and HCPs represented in our sample were reflected in the domains of an existing proposed survey instrument, the CQRA-PREM, a rheumatology-specific PREM originating in the United Kingdom. This instrument has been validated post publication for use with other rheumatic conditions [8], and translated into and validated in Dutch [9]. We have now undertaken this process with an Australian cohort.
The candidate instrument was first presented to participants in individual patient interviews (n=8). The purpose of individual interviews was to check the language, relevance, comprehensiveness, and completion time of the original CQRA-PREM, the latter as a measure of feasibility [10, 11]. Interviews were audio recorded, transcribed and content analysis was performed to identify recurring reflections on the language and comprehensiveness of the original instrument questions. Proposed changes to the original instrument were discussed among the research team and consensus was achieved. The modified instrument was thereafter referred to as the Australian-CQRA-PREM (ACQRAPREM).
The validation project was conducted in collaboration with the Australian Rheumatology Association Database (ARAD), a voluntary national database established to collect longitudinal outcome data from patients with inflammatory arthritis, administered by the Australian Rheumatology Association (ARA). Individual registrants of ARAD over 18 years of age, active in completing an unrelated online ARAD questionnaire within the preceding 12 months, and currently attending rheumatologist care, were invited by email to participate voluntarily in online survey data collection in September 2022. Participants who responded to the initial survey were invited to re-test at 6 months in March 2023. Participants completed the proposed ACQRA-PREM, a disease activity index relevant to their rheumatological diagnosis (Routine Assessment of Patient Index Data-3 for patients with Rheumatoid Arthritis, or Bath Ankylosing Spondylitis Disease Activity Index for patients with Ankylosing Spondylitis or Psoriatic Arthritis), a quality of life index (Assessment of Quality of Life score-6D), demographic survey items, and responses were linked to a comorbidity index (Rheumatic Diseases Comorbidity Index). Statistical analyses performed included exploratory factor analysis (EFA) and tests of reliability, validity, floor and ceiling effects, and minimum significant change value for the ACQRA-PREM instrument. Electronically signed informed consent for participation was obtained. The study was approved by the Central Adelaide Local Health Network Human Ethics Committee (reference 16836).
Results: The survey response rate was 707/1124 (63%) at the first data point (0 months) in September 2022. 459 (65%] respondents had a diagnosis of Rheumatoid Arthritis, 134 (19%) Psoriatic Arthritis, and 114 (16%) Ankylosing Spondylitis. The majority of respondents were female (n= 473, 67%), mean age was 62 years (SD= 11), mean disease duration 22 years (SD= 12). There were no significant differences in disease diagnosis, gender, age, disease duration between survey responders and non-responders. Median completion time (feasibility) of ACQRA-PREM was 299 seconds (IQR 130). Exploratory factor analysis extracted 5 factors, with all items loading similarly onto factor 1, indicating validity of an overall score for the ACQRA-PREM, averaged across all items. Remaining factors broadly recapitulated the domains of the original instrument. Cronbach’s alpha scores were > 0.8 for all factors identified by EFA, indicating internal consistency. Cronbach’s alpha for an average score of all item responses was 0.948, indicating reliability of the average score as an overall measure of the patient’s reported experience. The partial correlation between ACQRA-PREM score and standardised disease activity was not significant (rho=0.03, p=0.45), indicating divergent validity. There was no floor or ceiling effect for the average score. For the 6 month datapoint in April 2023, the survey response rate was 530/707 (78%). The test-retest reliability of the averaged PREM score, estimated by the intraclass correlation coefficient from a 2-way random effects model (Fleiss ICC [2,1]) was 0.74 (95% CI 0.70, 0.77). The ICC ranges from 0,1, and this value represented adequate reliability. Bland-Altman analysis was performed to investigate the agreement between measurements. Mean difference 0.00 indicated no systematic bias between measurement occasions. The 95% limits of agreement suggested that a difference in the mean score of 0.85 may be the least significant change for the ACQRA-PREM for an individual patient.
In summary, this study has field-tested the ACRQA-PREM, a modified rheumatology-specific PREM, with a large population of Australian patients, and has confirmed it as a valid and reliable instrument to measure self-reported care experience this cohort. As a novel application not previously reported, we have also demonstrated the utility of an overall, averaged PREM score for this instrument, which we propose can be used to follow patient reported experience longitudinally.
Future work: Following submission for publication, it is anticipated that the ACQRA-PREM will be distributed for use by rheumatology units around Australia. We intend to analyse the implementation process and report on the findings of the first data tranches from routine capture of experience data.
This work has been presented or will be presented at the following forums:
- Poster presentation, University of Adelaide Florey Postgraduate Research Conference, Adelaide, 2 August 2023.
- Future: Accepted abstract, Basil Hetzel Institute Research Expo, Adelaide, 19 Oct 2023.
This work will be submitted for consideration of presentation at:
- Future: Australian Rheumatology Association South Australia Annual Scientific Meeting, Adelaide, 27 Oct 2023.
- Future: Australian Rheumatology Association Annual Scientific Meeting, Christchurch, May 2024.
Once again we express sincere gratitude to Arthritis Australia for funding this important work.
PLAIN LANGUAGE REPORT
Our research team has a particular interest in understanding the experience of patients who attend rheumatology clinics around Australia. We acknowledge that people with rheumatic diseases are frequent, long-term attenders of healthcare services, that patients are expert consumers, and that they have vast experiential knowledge. To this end, understanding their care experiences is central to improving our healthcare services. To do this, we need a purpose-built survey to gather information from patients: a Patient Reported Experience Measure (PREM). We already collect information from patients about issues such as symptoms and test results, usually using surveys called Patient Reported Outcome Measures (PROMs), and have many of these surveys at our disposal. However, in the past we have lacked a PREM which is fit for use with Australian rheumatology patients. This is a gap in service delivery which we have sought to fill by undertaking this research.
To gather background information, we first met directly with patients to discuss their current experience of attending clinics, addressing both positive aspects and perceived shortcomings. We also engaged rheumatology clinicians to gauge opinion on what constitutes excellent care delivery. We found that patients’ experience was affected by issues such as waiting times and lack of care continuity, and that while clinicians are prioritising important issues such as information sharing and patient advocacy, often this is not clearly evident to patients, and change is needed to improve this. The findings of this study have been published in medical literature.
After analysing the priorities and concerns of both rheumatology patients and clinicians, we were satisfied that the types of issues raised would be adequately surveyed using an existing rheumatology PREM, but with modifications in the language and layout for Australian patients. We have performed the statistical work on the adapted PREM, called the ACQRAPREM, which stands for Australian-Commission for Quality in Rheumatoid Arthritis (CQRA)- PREM. The tests performed have ensured that the ACQRA-PREM asks the right questions and is reliable when used in the Australian context; in other words that it is fit for the intended use. This research reports on the testing and performance of our proposed PREM, for future use in rheumatology clinics across Australia. We anticipate that the findings of this study will be published in medical literature in the coming months.
We are hopeful that the ACQRA-PREM will be used by rheumatology units around Australia to collect this information from patients on a regular basis. We believe there is great potential to routinely capture experience-related data: to review which parts of care are performed well, and which parts need improvement. This information can be used directly to reduce barriers to providing great care and improve patient services.
References
- Bryant, M.J., et al., Joining forces to understand what matters most: qualitative insights into the patient experience of outpatient rheumatology care. Rheumatology Advances in Practice, 2023.
- Starks, Choose Your Method: A Comparison of Phenomenology, Discourse Analysis, and Grounded Theory. Qualitative Health Research (interlibrary loans also avaliable), 2007. 17(10): p. 1372-1380.
- Braun, V. and V. Clarke, Using thematic analysis in psychology. Qualitative Research in Psychology, 2006. 3: p. 77-101.
- Gale, N.K., et al., Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Medical Research Methodology, 2013. 13(1): p. 117.
- Australian Commission on Saftey and Quality in Healthcare., Consumer Fact Sheet 1: Introduction to the National Safety and Quality Health Service Standards. 2021.
- Care Quality Commission. The fundamental standards. 2022; Available from: https://www.cqc.org.uk/about-us/fundamental-standards.
- Institute of Medicine Committee on Quality of Health Care in America, in Crossing the Quality Chasm: A New Health System for the 21st Century. 2001, National Academies Press (US), Copyright 2001 by the National Academy of Sciences. All rights reserved.: Washington (DC).
- Bosworth, A., et al., Development and Validation of a Patient Reported Experience Measure (PREM) for Patients with Rheumatoid Arthritis (RA) and other Rheumatic Conditions. Current Rheumatology Reviews, 2015. 11(1): p. 1-7.
- Beckers, E., et al., Validation and implementation of a patient-reported experience measure for patients with rheumatoid arthritis and spondyloarthritis in the Netherlands. Clinical Rheumatology, 2020. 39(10): p. 2889-2897.
- Prinsen, C., et al., COSMIN guideline for systematic reviews of patient-reported outcome measures. Quality of Life Research, 2018. 27(5): p. 1147-1157.
- Terwee, C., et al., COSMIN methodology for evaluating the content validity of patientreported outcome measures: a Delphi study. Quality of Life Research, 2018. 27(5): p. 1159-1170.
Cellular Senescence as a Therapeutic Target for Knee Osteoarthritis
| Funded by: | Arthritis Australia & Arthritis South Australia |
| Recipient: | Dr Julia Kuliwaba |
| Intended Department | The University of Adelaide, Adelaide Medical School |
| Project: | Cellular Senescence as a Therapeutic Target for Knee Osteoarthritis |
Scientific summary:
Osteoarthritis is one of the most common, costly and disabling chronic conditions in Australia. One in five Australians aged over 45 years have osteoarthritis, with the prevalence increasing sharply with age. This musculoskeletal condition constitutes a major public health problem. The current failure to understand the underlying mechanisms of osteoarthritis has prevented the development of effective disease modifying treatments.
Of the many risk factors for osteoarthritis, the most important factor is chronological age. However, our understanding of the fundamental mechanisms, by which ageing contributes to osteoarthritis, is very limited. An emerging hypothesis implicates a causative role for cellular senescence (a modifiable ageing mechanism) in the development of osteoarthritis, with studies in mice showing its role in cartilage breakdown and loss. Senescence is a process by which a cell ages and permanently stops dividing but does not die. Over time, large numbers of these ‘zombie’ or senescent cells can build up in tissues throughout the body. These cells remain active and secrete pro-inflammatory cytokines, chemokines, and extracellular matrix proteins that can lead to inflammation and damage to nearby healthy cells. A critical knowledge gap is the role of senescence in the bone underlying cartilage (‘subchondral bone’); this bone plays an early and key role in osteoarthritis joint degeneration. Furthermore, it is unknown which specific cell types become senescent in the human ageing and osteoarthritis diseased bone microenvironment.
Using a tissue-based analytical approach involving specialised histochemical staining methods (eg. Sudan Black B staining optimised for decalcified, paraffin-embedded human bone tissue), we have identified, spatially localised and quantified the cellular types that are senescent in the tibial cartilage-subchondral bone microenvironment of the ageing human knee joint with osteoarthritis. The predominant cell types in the cartilage-subchondral bone tissue showing evidence of cytoplasmic lipofuscin accumulation (which is a cellular hallmark of senescence) are bone marrow myeloid cells, bone osteocytes, and non-calcified and calcified cartilage chondrocytes. Surprisingly, for myeloid and osteocyte cells, the percentage of cells that are senescent is very low, ranging from 2% to 5%. Increased numbers of senescent myeloid and osteocyte cells associate with more severe cartilage degeneration, but a larger study is needed to confirm this relationship. Senescent chondrocytes are mostly found in close proximity to fibrillated cartilage and are found associated with chondrocyte clusters, consistent with previous reports. Tibial cartilage-subchondral bone tissue from human knee joints without osteoarthritis also show very low numbers of senescent cells (<2%) in the bone marrow and bone tissue (i.e. osteocytes), but with no evidence of chondrocyte senescence. To investigate the functional significance of senescent cell presence in the subchondral bone and overlying cartilage in knee osteoarthritis, we will use human osteochondral explant cultures to assess bone and cartilage tissue response to senescent cell elimination or functional modulation. The osteochondral explant model system is currently being developed and optimised for these pre-clinical studies.
Preliminary study outcomes have been presented at invited symposia at the 2022 Annual Meeting of the Japanese Society for Bone Morphometry, and the European Calcified Tissue Society 2023 Annual Congress.
A current challenge in the ageing field is the lack of in vivo biomarkers of senescence specific for human tissues affected by chronic disease. Our future research goal is to discover novel senescence-associated proteins (i.e. novel biomarkers of senescence) specific for human osteochondral tissue, and validate whether senescence burden in joint tissues reflects senescence burden in synovial joint fluid and/or serum for patients with knee osteoarthritis. This is important, as an in vivo biomarker(s) of senescence specific for tissue changes associated with osteoarthritis disease severity, can be used to identify a minimal threshold or level of cellular senescence, above which an individual is more likely to benefit from senotherapeutic treatment (i.e. senescent cell clearing or senescent cell activity suppression therapies), in addition to monitoring changes in joint tissue senescence burden during clinical trials.
Bone Marrow Adipose Tissue in the Osteoporotic Bone
| Funded by: | Arthritis & Osteoporosis Western Australia & Arthritis Australia |
| Recipient: | Dr Kai Chen |
| Intended Department | University of Western Australia, School of Molecular Sciences |
| Project: | Bone Marrow Adipose Tissue in the Osteoporotic Bone |
Scientific summary:
Despite its static appearance, bone is a dynamic organ that continuously undergoes energy-intensive processes including osteoblastic bone formation and osteoclastic bone resorption (bone remodelling). The skeleton receives up to 10% of overall cardiac output which consumes a remarkable amount of daily energy to maintain bone homeostasis. Derangements of energy metabolism could therefore conceivably underpin bone cell dysfunction as observed in metabolic disorders of bone like osteoporosis. Osteoporosis is the most common bone disorder that has a high risk of potentially devastating fractures. One of the major pathological changes in the osteoporotic bone is the dramatic expansion of bone marrow adipose tissue (BMAT), which is inversely correlated with bone density and quality. BMAT has been recently shown to have an altered glucose metabolism, exhibiting significantly higher basal glucose uptake when compared to the axial bone. This implied a “derailed” glucose allocation during the pathogenesis of osteoporosis: Bone marrow adipocytes (BMAds) may significantly outcompete for glucose utilization with osteoblasts, thus leading to the energy-deficiency status for bone-forming osteoblasts and subsequent low bone mass. Hence, we hypothesised that de novo lipogenesis from glucose may also contribute to the pronounced expansion of BMAT. This has been a prospect in the field but has yet been proven due to technical inability.
To test our hypothesis, stable isotope-labelled glucose (13C-glucose, Cambridge Isotope Laboratories) were added to the primary bone marrow mesenchymal stem cells, followed by the stimulation of adipocyte differentiation. Following lipid droplet formation, cells were fixed and processed for correlative electron and ion microscopy (CEIM) analysis. Electron microscopy (EM) provides high-resolution morphologic information of the lipid droplets within BMAds, at a single organelle resolution. Ion microscopy (IM) using the NanoSIMS 50L makes it possible to record isotopic distributions with ~40-nm lateral resolution. The NanoSIMS instrument uses a focused caesium ion beam to bombard a section of tissue, releasing secondary ions (e.g., 12C-, 13C-). Images of cells are created from these secondary ions by mass spectrometry, which makes it possible to visualise the chemical elements of the biological samples. We used 13C/12C ratio-metric images to indicate the distributions of glucose in cells. Our results revealed that glucose was dramatically incorporated into lipid droplets during BMAd formation. To mapping this phenomenon in vivo, young (4-month-old, male) and ageing mice (18-month-old) were orally given with stable isotope-labelled 13C-glucose, in which the glucose was metabolically incorporated into the cells or tissues. Following mice euthanasia, the tibiae were collected and processed for CEIM analysis. We showed that the signal of glucose (13C/12C ratio) contributes to the BMAT formation, as well as BMAT expansion in the ageing-induced osteoporotic bone. Taken together, our study provided the direct evidence that the glucose utilization significantly contributes to de novo lipogenesis within the bone marrow.
Based on these findings, we reasoned that the excessive de novo lipogenesis of glucose could be a crucial event during the expansion of BMAT in the osteoporotic bone. In the future, we will further investigate the mechanisms underlying the derailed glucose allocations between bone cells and BMAds, thus developing therapeutic solutions to osteoporosis by reducing the metabolic incorporations of glucose into the lipid droplets but increasing its utilisation as energy substrate for osteoblasts.
Lay Summary:
Bones may seem static, but they are actually dynamic organs that continuously go through processes like bone formation and bone resorption (remodelling). To maintain bone health, the skeleton requires a significant amount of dietary nutrients. Problems with how the body uses energy could lead to bone disorders like osteoporosis, which makes bones weak and prone to fractures. In osteoporosis, one major feature is the increase in bone marrow adipose tissue (BMAT), which is fat in the bone marrow. This increase is linked to lower bone density and quality. Recently, it was found that BMAT has altered glucose metabolism, meaning it uses more glucose than the bone itself. This “misallocation” of glucose might be responsible for the lack of energy for bone-forming cells (osteoblasts) and the subsequent reduction in bone mass.
To test this hypothesis, we used a labelled form of glucose (13C-glucose) and added it to bone marrow cells to see how BMAT formed. We developed advanced imaging techniques (electron and ion microscopy) to study the process. The results showed that glucose was significantly incorporated into fat droplets during the formation of bone marrow fat. We also conducted similar tests in living mice and found that glucose played a role in BMAT formation in the aging-induced osteoporotic bone.
Based on these findings, it appears that excessive glucose processing into fat could be a crucial factor in the expansion of BMAT in osteoporotic bones. We hope to investigate further to understand why this glucose misallocation occurs and find ways to develop treatments for osteoporosis. One potential approach is to reduce the conversion of glucose into fat while promoting its use as an energy source for bone-forming cells.
In summary, this study provides direct evidence that glucose utilisation significantly contributes to the formation of fat within the bone marrow, which may help us develop better therapies for osteoporosis in the future.
Breathlessness in Systemic Sclerosis
| Funded by: | ARA Victoria |
| Recipient: | Dr Laura Ross |
| Intended Department | St Vincent’s Hospital Melbourne, Department of Rheumatology |
| Project: | Breathlessness in systemic sclerosis |
Scientific report: The award of this fellowship has enabled me to establish a post-doctoral research program investigating the causes of physical disability and poor exercise tolerance in systemic sclerosis. Over the period of fellowship funding, I have published seminal studies that describe the burden of exercise impairment in systemic sclerosis, with the results of this study linking the burden of myocardial fibrosis (detected by cardiac MRI) and exercise tolerance in systemic sclerosis for the first time (Ross et al Seminar Arthritis Rheum 2023). The results of this study challenge the paradigm of so-called ‘subclinical’ cardiac involvement in systemic sclerosis, as I was able to demonstrate the physiological importance of diffuse myocardial fibrosis even in the absence of specific cardiac symptoms. Using results from cardiac and skeletal muscle MRI, respiratory function testing, echocardiography, and cardiopulmonary exercise testing, I have been able to highlight the profound extent of exercise intolerance in systemic sclerosis, as well as the multi-system disease specific contributors to physical impairment. I have performed additional studies to further investigate the multi-organ involvement causing breathlessness, evaluating each of left ventricular diastolic dysfunction, pulmonary involvement, and skeletal muscle involvement in systemic sclerosis.
As a post-doctoral fellow, I have supervised a PhD student (Fairley, commenced 2022) and three rheumatology advanced trainees (Shah, Moxey, Strathmore), resulting in two accepted manuscripts, an oral conference presentation (ARA ASM 2023) and a further 5 manuscripts currently in preparation for submission.
The Arthritis Australia Fellowship has permitted me to build my track record, such that I was able to submit NHMRC Investigator Grant and MRFF Early Career Research Grant Applications in 2023, with the results of these applications still pending. This future funding is intended to support my ongoing interest in the investigation of the causes of physical disability and breathlessness in systemic sclerosis by leading a fully-powered randomised controlled trial of exercise therapy as an adjunct to standard of care in the management of systemic sclerosis.
A list of the manuscripts, conference presentations, and prizes awarded over the funding period are listed below:
Original research publications
- Fairley JL, Ross L, Quinlivan A, et Sudden cardiac death, arrhythmias and abnormal electrocardiography in systemic sclerosis: A systematic review and meta-analysis Semin Arthritis Rheum 62 : 152229
- Fairley JL, Hansen D, Ross L, et al. Clinical characteristics and survival of pulmonary arterial hypertension with or without interstitial lung disease in systemic sclerosis Arthritis Res Ther 2023 25 (1) 77
- Ross L, McKelvie P, Reardon K et al Muscle biopsy practices in the evaluation of neuromuscular disease: A systematic literature review Neuropath Appl Neurobiol 2023 in press
- Ross L, Costello B, Lindqvist A et al Disease specific determinants of cardiopulmonary fitness in systemic sclerosis, Semin Arthritis Rheum 2023 Feb 58:152137
- Ross L, Proudman S, Walker J et al Evaluation of patient and physician assessments of gastrointestinal disease activity in systemic sclerosis J Rheumatol 2022 Nov 15 doi: 3899.jrheum.220832 online ahead of print
- Ross L, Nikpour M, D’Aoust J, Khanna D, Merkel PA, Pauling JD, Baron M Patient and physician global assessments of disease status in systemic sclerosis, Arthritis Care Res (Hoboken) 2022 epub Nov 7, online ahead of print
- Ross L, Patel S, Stevens W et al The clinical implications of left ventricular diastolic dysfunction in systemic sclerosis, Clin Exp Rheumatol 2022 40(10) 1986-1992
- Ross L, Lindqvist A, Costello B et al Using MRI to map the hidden burden of muscle involvement in systemic sclerosis Arthritis Res Ther 2022 24:84
- Ross L, Costello B, Brown Z et al Myocardial fibrosis and arrhythmic burden in systemic sclerosis
Rheumatology (Oxford) 2022 61(11) 4497-4502
Conference presentations
- EULAR Congress, Milan 2023, ‘Development and validation of the novel Scleroderma Clinical Trials Consortium Activity Index’ (Oral abstract presentation)
- Australian Rheumatology Association Annual Scientific Meeting, Hobart Australia 2023
‘Musculoskeletal manifestations of systemic sclerosis’ (Invited oral presentation)
- Australian Rheumatology Association Annual Scientific Meeting, Perth Australia 2023 ‘Characterising Exercise Capacity in Systemic Sclerosis Using Cardiac Magnetic Resonance Imaging, Skeletal Muscle Imaging and Cardiopulmonary Exercise Testing (Oral abstract presentation)
- American College of Rheumatology Annual Scientific Meeting, Philadelphia USA, 2022 ‘Cardiac Involvement in Systemic Sclerosis: The Heart of the Matter’ (Invited oral presentation)
- Inaugural Cardiometabolic Collaborative Symposium, Melbourne Australia, 2022 ‘Physical disability in scleroderma – how big a problem and what are the causes?’ (Invited oral presentation)
Prizes
- TJ Martin Medal – Best PhD thesis passed at Eastern Hill Campus, University of Melbourne (2023)
- EULAR Congress Abstract Prize – Clinical Sciences (2023)
- Australian Rheumatology Association Early Career Research Excellence Award (2023)
- University of Melbourne MDHS Dean’s Award for Excellence in Graduate Research (2022)
- Australian Rheumatology Association Best New Investigator Award (2022)
- Aikenhead Centre for Medical Discovery Research Week Best Poster – Clinical Sciences (2022)
Plain language summary (Lay report): Systemic sclerosis (SSc), or scleroderma, is a devastating autoimmune disease often affecting young and middle-aged adults, that causes scarring or fibrosis of the skin and internal organs. Feeling breathless is very commonly reported by patients, and routine tests available as part of usual clinical care often fail to identify a cause of these symptoms. It is well known that SSc affects the lungs and blood vessels leading to pulmonary hypertension (high blood pressure in the vessels connecting the heart and lungs). However, we now understand that SSc causes scarring in the heart muscle and can affect the skeletal muscles even in the absence of clear symptoms. Using cardiopulmonary exercise testing (CPET), I have been able to show that 50% of patients with SSc have a sufficiently impaired exercise capacity that day-to-day tasks are challenging, and they are likely to require assistance. Changes that occur in patients’ heart, lungs and skeletal muscles all contribute more breathlessness and reduced exercise tolerance. Future work is needed to better understand the specific mechanisms of exercise intolerance and whether treatment strategies such as a targeted, prescribed exercise program can help ameliorate symptoms of breathlessness in patients with SSc.
I would like to thank Arthritis Australia for their generous support of me as an early post-doctoral researcher. The award of this fellowship has been integral to my being able to continue my research career following on from my PhD. I hope that the results of my studies will further inform our understanding of exercise impairment in systemic sclerosis and that these results will serve as an effective rationale for acquiring future funding to support a fully-powered disease-specific exercise intervention study.
Linking the presence of live ileal-derived bacteria in the joint as a triggering factor for the development of spondyloarthropathy
| Funded by: | ARA Research Fund |
| Recipient: | Professor Ranjeny Thomas |
| Intended Department | University of Queensland Diamantina Institute |
| Project: | Linking the presence of live ileal-derived bacteria in the joint as a triggering factor for the development of spondyloarthropathy |
Lay Summary
Spondyloarthropathy (SpA) is a family of diseases, which include ankylosing spondylitis, psoriatic arthritis, and reactive arthritis that usually affect the spine and the large joints of the arms and legs. It has also been associated with inflammation in the eyes, skin and intestines, representing a major burden for patients. Current treatments aim to relieve pain and stiffness, prevent or delay disease progression and additional complications and spinal deformity. Treatment is lifelong. Biologic disease-modifying drugs, such as tumor necrosis factor (TNF) blockers or interleukin (IL)-17 inhibitors do not suppress all symptoms. Patients may have to stop therapy due to adverse events or loss of efficacy. Since AS has no cure, there is an urgent need for new therapeutic options for this important healthcare issue. Treating the cause of the disease rather than the symptoms would be a major advance in patient care.
Similar to patients with inflammatory bowel disease, more than half of SpA patients have intestinal inflammation and increased permeability. Clinical observations and animal studies in other forms of arthritis suggest that this permeability could facilitate leakage of bacteria and other microbes normally residing in gut to the circulation, and then possibly to sites of inflammation such as joints, eyes and skin. This is however an unresolved question, especially in SpA. Using a mouse model of SpA where a single gut bacterium is introduced, we showed that this gut-derived bacterium can be carried by immune cells to the joints and there drive arthritis. Overall, this confirms the link between the gut and the development of arthritis, which opens exciting new opportunities to develop curative strategies to enhance gut health and control harmful bacteria. How gut bacteria in the joint activate arthritogenic inflammation is still unknown but is the subject of this continuing project in Prof. Thomas laboratory, in order to get closer to developing novel antigen-specific immunotherapy for SpA.
Scientific Summary
Ankylosing Spondylitis (AS) is a form of chronic inflammatory arthritis of the spine and peripheral joints. It is closely related to other spondyloarthopathies (SpA), such as reactive arthritis triggered by bacterial infections and arthritis associated with inflammatory bowel disease (IBD). IL-23 dependent spondylitis, arthritis and ileitis recapitulate human SpA in curdlan-treated SKG mice. SpA patients and SKG mice have fecal dysbiosis with increased gram-negative bacteria. In SKG mice, anti-IL-23 treatment supports homeostatic bacteria, gut integrity and is sufficient to prevent arthritis. In this project, germ-free (GF) SKG or control BALB/c mice were colonised for 4 weeks with Parabacteroides sp. (Para), Lactobacillus murinus (Lm), or altered Schaedler flora (ASF) comprising Para, Lm, Mucispirillum Schaedleri, and Clostridium sp., then given curdlan. We assessed gene transcripts, goblet cells and bacterial DNA in small intestine (ileum) and in the inflamed joints (rear paw). Para or Lm monocolonised SKG mice, but not GF mice or BALB/c mice, developed IL-23-dependent ileitis and arthritis within five weeks post-curdlan. One week post-curdlan, in the ileum, stress-related Il23a, Grp78 and sXbp1 gene expression was significantly increased, and the number and tight junctions of mucin-producing goblet cells were compromised, confirming reduced gut barrier integrity. Concomitantly, Para or Lm translocated from gut lumen to the villi where macrophages and neutrophils captured them. Bacterial DNA was found within macrophages and neutrophils in inflamed joints. In contrast, SKG colonised with ASF were protected from ileitis but not arthritis, with enhanced regulatory mechanisms in the gut. Overall, we showed that gut-derived bacteria are a key player in the development of arthritis. This opens exciting new opportunities to treat the cause of the disease rather than the symptoms, by developing curative strategies to enhance gut health and control harmful bacteria.
How gut bacteria in the joint activate arthritogenic inflammation is still unknown. This is the subject of ongoing work in Prof. Thomas laboratory. Recently, expanded CD8+ T cell clones were identified by a UK team in blood, joints and eye of multiple AS patients. The clones recognised peptides derived from gut bacteria and self-antigens presented by HLA-B27. Our laboratory is examining expanded CD8+ T cell clonotypes in gut and synovial tissue of HLA-B27+ AS patients with arthritis with and without ileal inflammation to understand which T cells are pathogenic in AS, what they recognise, where they migrate, how they signal and how they differ during active disease, remission and the healthy state. Ultimately we aim to understand disease-relevant antigens in order to develop antigen-specific immunotherapy for SpA.
Evaluating and targeting unhelpful beliefs about osteoarthritis and activity in those with painful osteoarthritis
| Funded by: | Arthritis Australia |
| Recipient: | Dr Brian Pulling |
| Intended Department | Body in Mind Research Group |
| Project: | Evaluating and targeting unhelpful beliefs about osteoarthritis and activity in those with painful osteoarthritis |
Plain language summary
Research Overview:
With the support of Arthritis Australia, I have developed two tools that can be used by researchers and clinicians to understand how people with knee osteoarthritis think about their condition. I was also able to use complex analysis methods (e.g., network analysis) to further explore our data and gain a greater understanding of people’s complex beliefs about their condition. This will allow researchers and clinicians in the future to conduct more advanced research, testing new treatments for knee pain.
What questions did the grant set out to answer? What problems did you try to solve, or gaps in knowledge did you try to fill? Why is this important?
I set out to create assessment tools that would help researchers and clinicians better understand the complex experience of knee pain. Existing tools (such as questionnaires) try to simplify this complex experience. This is a problem, because we know the way people experience osteoarthritis is based on the many factors that all contribute to the way in which they think about their condition. Not only that, but these assessments only evaluate how we “explicitly” think, but research has shown that the way we unconsciously think about these issues (our “implicit” attitudes) is also important to evaluate. This work is (to our knowledge) the first to evaluate the complex network of knowledge, beliefs, and experiences that all impact a person’s experience of knee pain using both explicit and implicit methods.
What did you discover during the course of the grant?
We have discovered that people do indeed hold unhelpful beliefs about osteoarthritis – this was not surprising, however, we were also able to show that people with knee OA hold heightened implicit threat-activity attitudes (i.e., attitudes that physical activity is dangerous) relative to people without pain. Importantly, these implicit attitudes are not associated with explicit self-reported beliefs, suggesting that we are capturing something unique.
Project One: Development & evaluation of an OA attitudes/beliefs questionnaire
The questionnaire comprehensively evaluates a person’s beliefs about knee osteoarthritis. We found that there are factors that are critical to how people think about osteoarthritis named ‘Activity and Damage’, ‘Learning’, ‘Influences’, ‘Pain Science’, ‘Joint Improvement’. This questionnaire has good psychometrics, meaning that it performs well under statistical tests that evaluate its usefulness to researchers and clinicians. We expanded this project to use a method called “network analysis”. This method analyses a huge amount of data and visualises it, so that we can see how different concepts are related to one another. This is important to future research where we will attempt to target specific concepts when making education interventions about pain more effective.
Project Two: Development and evaluation of unconscious beliefs/attitudes towards movement
We found that people with knee pain implicitly associate rest with safety, and activity with danger. We found that this implicit association does not match well with what they explicitly believe about the safety or danger of activity. We found that people with pain have much stronger associations between activity and danger than people who do not have pain. Taken together, these findings suggest that implicit attitudes may differ from what people explicitly say. Sometimes, people say that “exercise is safe”, but implicitly they may feel that exercise may be dangerous. This is important to know, because past work has shown that these implicit attitudes can sometimes better predict behaviour than what people self-report. So, our findings might mean that even though people report they do not think activity is dangerous, having negative implicit attitudes may still reduce their activity levels.
Project Three: Understanding links between objective physical activity levels and beliefs in knee OA
Data collection for this project is tied in part to an ongoing clinical trial that will conclude in 2023. The assessments developed in projects one and two are instrumental in evaluating the complex links between physical activity and beliefs about knee OA. This project would not be possible if projects one and two were not successful.
Project Four: Pilot study evaluating a new educational intervention
This pilot study has not yet been undertaken. However, successful completion of the first three projects has made this project possible in the future. (Note: completion of this project was not an aim of the scholarship – it was provided as an example of future work).
Have the findings of the research already benefitted people with musculoskeletal disease? How might the findings inform further research to help sufferers in the future?
The purpose of this work was to develop the tools researchers and clinicians need in order study and develop better treatments for people with knee OA. Now that we have developed these tools and shown that they can be used to identify how people think about and experience osteoarthritis, we can move forward and use them to improve existing treatments for knee pain, and even develop new innovative treatments for this complex disease. There are already several projects underway that use these tools, and several more in the early planning stages.
| Project Title | First Author | Project Summary |
| EPIPHAKnee Trial | A/Prof Tasha Stanton (University of South Australia) | A large scale randomised controlled trial, to test a novel pain education treatment with a walking program for people with painful knee osteoarthritis. The questionnaire will be used to evaluate whether the treatment changes the way people think about their condition. |
| Neural contribution to pain and motor function in people with painful knee osteoarthritis | Johanna Paddick (University of South Australia) | A clinical study using transcranial magnetic stimulation to evaluate the neural processes underlying pain and function in OA. The questionnaire will be used to evaluate how people think about osteoarthritis. |
| Development of a clinician-version of the questionnaire and evaluate reliability and validity. | Prof Cormac Ryan (Teeside University) | This project aims to develop a version of the questionnaire that can be used to evaluate how clinicians think about osteoarthritis. This has important potential impacts on the way we teach future clinicians about osteoarthritis. |
| OA Champions: Pain Science Education Peer Mentorship Program | Monique Wilson (University of South Australia) | A pilot feasibility study of peer mentorship for people with knee osteoarthritis. |
| Impact of myth-busting videos on beliefs about osteoarthritis: ‘The Cartilage Story’ | Dr. Felicity Braithwaite | Evaluating ‘The Cartilage Story’: a video that targets the ‘wear and tear’ myth. The primary aim is to evaluate the video’s ability to influence beliefs about OA. |
Are you planning to continue the research?
I hope to be able to continue to pursue this important work as a post-doctoral researcher. The tools developed during this work are freely available online for researchers and clinicians to use as they continue their own work.
Scientific summary (Scientific report)
What were the main scientific objectives of the grant?
There were four aims while conducting this work:
1) to create a measure of a person’s explicit conceptual beliefs related to knee OA, so that pre-existing misconceptions can be comprehensively evaluated;
2) to create a measure of implicit (unconscious) conceptual beliefs, so that any differences between explicit reports and implicit biases can be explored;
3) to determine if beliefs (as evaluated by the new tools projects one and two) are associated with physical activity levels;
4) to facilitate conceptual change by using enhanced education methods, specifically creating dissonance by targeting those concepts that are important misconceptions of an individual’s understanding of their knee OA.
The first two of these have been successfully completed. Data collection related to the third objective is ongoing and will be complete in 2023. The fourth objective represents a path forward that will utilise the findings from the previous three projects (was not anticipated to be completed within the scholarship time-frame).
What were the main scientific achievements of the grant? Your answer should be at least 200 words.
After collecting data from more than 400 people with painful knee osteoarthritis, we created a measure of a person’s explicit conceptual beliefs (allowing evaluation of pre-existing misconceptions). Via factor analysis, we identified that this questionnaire assesses six major constructs related to osteoarthritis, named ‘Activity and Damage’, ‘Learning’, ‘Influences’, ‘Pain Science’, ‘Joint Improvement’, and ‘Complex Influences’. Additionally, we have also created a “conceptual framework” tool. This first of its kind tool uses network analysis to spatially map the way people think about osteoarthritis. This method visualises large datasets to show the multidimensional relationships between individual concepts. In doing so, educational curricula can be tailored to specifically target the educational gaps held by an individual, rather than systematised to the population at large. Use of our conceptual framework tool may facilitate more nuanced and individualised clinical osteoarthritis education.
After collecting data from 236 people with painful knee OA and another 218 healthy controls, we have created an online implicit association test (IAT) that assesses implicit conceptual attitudes surrounding threat and physical activity. We discovered that people with painful knee osteoarthritis hold unhelpful implicit attitudes toward physical activity; these threat-activity attitudes are heightened relative to those held by people without pain. Furthermore, we have shown that this implicit assessment task evaluates attitudes in a way that is not possible using explicit measures, suggesting that this task may provide additional benefit in predicting someone’s activity-related behaviour (i.e., in addition to explicit self-report). Importantly, we found that the IAT only weakly correlates with explicit assessments of belief about concepts related to pain knowledge, fear of movement, and beliefs about exercise. This suggests that the IAT evaluates a related but unique construct from that assessed by explicit measures. The IAT has good internal consistency and low error rate, further demonstrating the utility of the task for researchers and clinicians interested in evaluating implicit attitudes about threat and activity in people with knee OA.
What problems, if any, did you encounter in achieving the project’s objectives, and how did you address them?
The most challenging aspect of this work was recruitment, as all the analyses relied on large sample sizes with a diverse range of experiences and participant backgrounds. Progress with projects three and four were particularly impacted by the COVID-19 pandemic, and so data collection relevant to objective three is continuing via an NHMRC funded large scale randomised controlled trial (recruitment delayed).
Study four outlines a clear path for translation of the first three studies. The development of network analysis in Project two facilitates future research about the design and implementation of tailored educational curricula based on the unique conceptual framework held by a given individual.
Research Dissemination:
Three manuscripts related to this work have been submitted or are in the final stages of preparation for submission to scholarly journals.
Pulling BW, et al. (2023). Item development and pre-testing of an Osteoarthritis Conceptualisation Questionnaire to assess knowledge and beliefs in people with knee pain. Manuscript Accepted at PLoS One.
Pulling BW, et al. (2023). Osteoarthritis Conceptualisation Questionnaire (OACQ) Phase 2: Scale development with factor and network analyses. Manuscript in preparation for submission to Arthritis and Rheumatology.
Pulling BW, et al. (2023). Osteoarthritis Conceptualisation Questionnaire (OACQ) Phase 3: Rasch analysis and reliability assessment.
Pulling BW, et al. (2023). People with Painful Knee OA Implicitly Believe that Movement is Dangerous. Manuscript in preparation for submission to PAIN
Findings related to this work have been presented at national and international conferences (see abstracts attached).
Pulling BW et al., ‘Item Development and Pre-Testing of the Osteoarthritis Conceptualisation Scale (OACS) to Assess Knowledge and Beliefs in People with Knee Pain’. 2022 Australian Pain Society 42nd Annual Scientific Meeting. Hobart, Tasmania, Australia, 11-13 April 2022. Poster and rapid oral presentation.
Pulling BW et al., ‘KneeIAT: People with Painful Knee Osteoarthritis Implicitly Believe that Movement is Dangerous’. 2022 International Society for the Study of Pain Annual Scientific Meeting. Toronto, Canada. 19-24 September 2022. Poster (Accepted).
This work was presented at the Calvary Hospital Grand Rounds, on 26 May 2022.
I have also written two blogs that have been shared online and/or with organisation email newsletters (NOIjam.com; Pain Revolution Rev’d Up newsletter).
Is running bad for my knee? A study using novel MRI techniques to investigate knee health in young runners at high-risk of knee osteoarthritis.
| Funded by: | Arthritis Australia and Arthritis NSW |
| Recipient: | Dr Andrea Bruder |
| Intended Department | La Trobe Sport and Exercise, Medecine Research Centre – School of Allied Health, Human Services and Sport. |
| Project: | Is running bad for my knee? A study using novel MRI techniques to investigate knee health in young runners at high-risk of knee osteoarthritis. |
Plain Language Summary
What question did the grant set out to answer? What problems did you try to solve, or gaps in the knowledge did you try to fill? Why is this important?
Knee osteoarthritis (OA) is a painful and debilitating condition that develops in 50% of young Australians after knee surgery. In these individuals with a history of knee surgery, OA results in reduced function and poor quality of life before the age of 40 years, at a time when family and work responsibilities are high. One potential contributing factor to the faster development of OA is participation in high-impact exercise, such as running. Running, a form of physical activity critical for the health of all Australians, has many health-related benefits. This form of physical activity is popular due to its affordability and accessibility to people of all ages, and often taken up as an alternative to more high demanding sports such as football or netball. However, the common misconception is that running is bad for your knees. We currently do not know if running is a safe form of exercise for high-risk knee OA individuals (i.e. post-knee surgery).
Hence, this project set out to examine knee joint cartilage and bone (using novel- magnetic resonance imaging (MRI), and changes in knee pain and quality-of-life over one year in 50 high-risk runners (i.e. post-knee surgery). Critically, we wanted to fill the knowledge gap concerning the safety of running for high-risk young adults and explore how regular running might change knee OA symptoms. To do this, we set out to explore the association between running load (i.e. amount of running per week) on knee pain and quality of life in a high-risk running group.
Specifically, the grant enabled a group of 50 high-risk runners to undergo physical assessments, an MRI scan and questionnaires over 12-months. The knowledge obtained from this study will help runners, individuals at high-risk of future knee OA, and clinicians make informed decisions about running for health. This project will also provide a platform for future larger scale studies to follow-up beyond one year, and/or design studies to prevent the early development of knee OA in early and mid-adulthood.
What did you discover during the course of the grant?
We successfully recruited 50 runners (36 males, 14 females) with a history of knee surgery. On average, runners were aged 33 years, had a healthy body mass index and ran 4 times per week (total 32 kilometers per week). The preliminary findings from this grant helped us to understand that runners at high-risk of knee OA, report mild knee pain. Despite this, high-risk runners run regularly, and importantly this pain does not worsen over 12 months. Physical testing (i.e. knee extensor and knee flexor strength tests) allowed us to understand that knee strength of the operated leg was not related to knee symptoms at 12 months. Comparing these results to previously collected data among 50 runners without a history of knee surgery, there was no significant difference in the change of knee pain over 12 months. These findings suggest that regular running does not increase knee pain in the short-term, making it a physical activity option for individuals after knee surgery.
The MRI scans obtained at baseline will be analysed by a team of researchers trained to analyse MRI scans. We hypothesize that a number of runners with a history of knee surgery will demonstrate knee cartilage loss, but that these changes will not be associated with worsening knee OA symptoms at 12 months.
Once analysis of the completed MRI scans has been completed (end 2023), we will understand the prevalence of knee knee joint cartilage and bone among runners at risk of high risk of knee OA, and be able to explore if these features are associated with knee pain at 12 months.
Have the findings of the research already benefitted people with musculoskeletal disease?
The runners at high-risk of knee OA have benefited greatly from the follow-up assessments. Each runner was assessed by a physiotherapist and provided their individual results at each assessment. The physical testing gave runners an insight into the current level of strength and function of their operated limb compared to their non-operated limb. Their results were interpreted compared to uninjured group averages. Where operated limb assessment results were below uninjured averages, this has often prompted them to continue working on their physical functioning to maximise levels of activity. This is important, as there is good evidence for non-surgical management (strengthening and activity modification) for knee OA. Each participant received a copy of their MRI report written by a clinical radiologist.
To enhance project relevance and engagement with the running community, we recruited two project ambassadors, an Olympian marathon runner (Ellie Pashley) and a runner with a history of knee surgery (Tyler Scarce) who have a combined social media following of >8,000 followers. With their help, we established a project podcast (https://trail.latrobe.edu.au/category/podcast/) and blog (https://trail.latrobe.edu.au/blog/) to promote recruitment, improve running-related knowledge and disseminate study findings within the running community. For example, our broad work in this space has motivated a post-graduate student within our team to create a layperson infographic busting the myth that running causes OA http://dx.doi.org/10.1136/bjsports-2021-104342. Our podcast features researchers, coaches, runners and OA experts. We have also created a project social media instagram account (handle: @trail_running_study) where we communicate study updates and findings.
Presentation of the research project and findings have begun at conferences (2 in 2022 at the Australian Sports Medicine Association Conference) and in journal publications. We will continue to present at various sports medicine, physiotherapy and rheumatology conferences, sports medicine clinics, and disseminated via social media platforms. This will enable researchers, and clinicians, to gain important insights about running and knee outcomes.
Finally, this grant has enabled our research group to collaborate with international running researchers. Our research group is coordinating an international workshop on running and knee health alongside the World Congress of OA in Denver, USA on 17th March 2023.
How might the findings inform further research?
The pilot data from this grant has enabled us to successful gain additional funding, >$60,000 across two La Trobe University grant schemes to: 1) increase recruitments numbers to reach 100 runners with, and 100 runners without a history of knee surgery, 2) recruit additional
women runners to achieve sex and gender parity and explore women-specific factors (e.g., menstrual cycle, caring roles) on future knee OA outcomes, 3) long-term follow-up at 4 years, and, 4) explore inflammatory blood biomarkers among high-risk runners.
We have already commenced recruiting additional runners with a history of knee surgery to undergo physical assessments, MRIs, questionnaires, and blood collection. The increased sample size will allow us to perform sufficiently powered analyses of the preliminary findings enabled by this grant.
Re-assessment at 4 years (enabled by new grants) will allow us to understand the longer-term trajectory of knee OA structure and pain among runners with and without a history of knee surgery. If we know which runners, and which factors after knee-surgery are likely to have deteriorating pain and quality of life, we can design interventions that can prevent or slow knee OA progression. Alternatively, if we find that running behaviour has no effect on the long-term trajectory of knee pain, cartilage or bone we will advance our knowledge about the health effects of running and can encourage it as a safe form of exercise despite its high-impact. The outcomes will empower and support those with, or at risk of developing, knee OA to make informed decisions about sport participation for future health and well-being.
SCIENTIFIC REPORT
BACKGROUND
Knee osteoarthritis (OA) is a painful and debilitating condition that develops in 50% of young Australians after knee surgery (1, 2). In these individuals with a history of knee surgery, they will often experience persistent pain, functional loss and poor quality of life before 40 years (3). Knee OA has a significant individual and societal burden, often amplified by co-existing parental and occupational responsibilities.
One potential contributing factor to the faster development of OA is participation in high-impact exercise, such as running. Running is popular due to its affordability and accessibility to people of all ages, and often taken up as an alternative to contact/twisting sports such as football or netball. However, there is a general perception that running is bad for your knees (4) due to the high-impact joint loads associated with running (5, 6). Systematic reviews have not established a causal relationship between running and OA risk, suggesting that healthy joints can tolerate running loads. For individuals already susceptible to OA (i.e. following knww surgery), the impact of regular running on knee joint health and associated symptoms is unknown. Mediating factors such as lower limb strength may dissipate knee joint loads, to minimise radiographic and symptomatic OA development in young adults following knee surgery (7). Since running confers many health benefits and likely to be a physical activity of choice after knee surgery, it is vital to establish if running loads are associated with the presence of early OA and its symptoms trajectory in a high-risk population (i.e. post-knee surgery).
MAIN SCIENTIFIC OBJECTIVES OF THIS GRANT
1) To quantify knee OA structural features and symptoms in 50 runners with a history of knee surgery
2) Explore relationship of lower limb strength on knee OA symptoms in runners with a history of knee surgery at 12-months
3) To compare knee OA symptom trajectory over 12 months between runners with a history of knee surgery to previously collected data in runners without a history of knee surgery
4) Investigate the relationship between running behaviour on symptom trajectory over 12 months in runners with a history of knee surgery.
SCIENTIFIC ACHIEVEMENTS OF THE GRANT
This grant is enabling us to explore knee OA structural features and symptomatic trajectory in 50 runners at high-risk of OA (i.e. post-knee surgery). We recruited 50 runners (36 men, 14 women) and collected knee MRIs, assessed knee flexor and extensor strength in our laboratory, and knee OA symptoms (at baseline and 12-months).
This project has provided preliminary evidence that people with a high risk of OA who regularly run, do not have a meaningful worsening of knee OA symptoms after 1 year. The findings contribute to our knowledge about the health effects of running providing preliminary evidence that running may not be bad for your knees. Combined with additional data, these outcomes may empower and support those with, or at risk of developing, knee OA to make informed decisions about sport participation choice and its association with future health and well-being outcomes.
The participant characteristics at baseline laboratory testing are presented in Table 1. On average, runners were aged 33 years, healthy body mass index and ran 4 times per week (total 32 kilometres per week). At baseline testing, high-risk runners self-reported mild knee OA symptoms on the Knee injury and Osteoarthritis Outcome Score (KOOS) (mean=85 (SD 12.5) out of 100, 0 = extreme knee problems, 100 = no knee problems). Whilst mild, these self-reported symptoms were significantly worse compared to runners without knee surgery (difference -8.42 (95%CI -13.00 to -3.84) points worse (p < 0.001).
Figure 1 presents the change in KOOS scores at baseline and at 12-months between runners with and without a history of knee surgery. Data from four of the five KOOS subscales are reported together with a common composite measure – KOOS4 (the average of four of the five subscale of the KOOS covering pain, symptoms, difficulty with sport and recreational activities and quality of life). Although runners without a history of knee surgery generally reported less knee symptoms, there were no clinically meaningful changes in knee symptoms between the two groups (Table 2).
In relation to modifiable risk factors,
– performance on knee extensor muscle strength testing at baseline following knee surgery was not associated with any KOOS symptoms at 12 months (results ranged from KOOS pain -0.013, P= 0.608 to KOOS sport and recreation 0.053, P= 0.291.
– performance on knee flexor muscle strength testing at baseline following knee surgery was not associated with any KOOS symptoms at 12 months (results ranged from KOOS quality of life -0.033, P= 0.713 to KOOS sport and recreation 0.112, P= 0.192).
When exploring relationships between running behaviour and future knee OA symptoms, preliminary results suggest no relationship between any training load variable and future knee OA symptoms. Specifically, no relationship between future knee OA on number of runs per week (-0.58, 95%CI -1.987 to 0.830, P=0.412), total distance per week (-0.05, 95%CI -0.16, 0.07, P=0.435), longest single run in a session of an 8-week period (-0.165, 95%CI -0.45 to 0.11, P=0.246) and highest weekly load in an 8-week period (-0.05, 95%CI -0.14, 0.05, P=0.309).
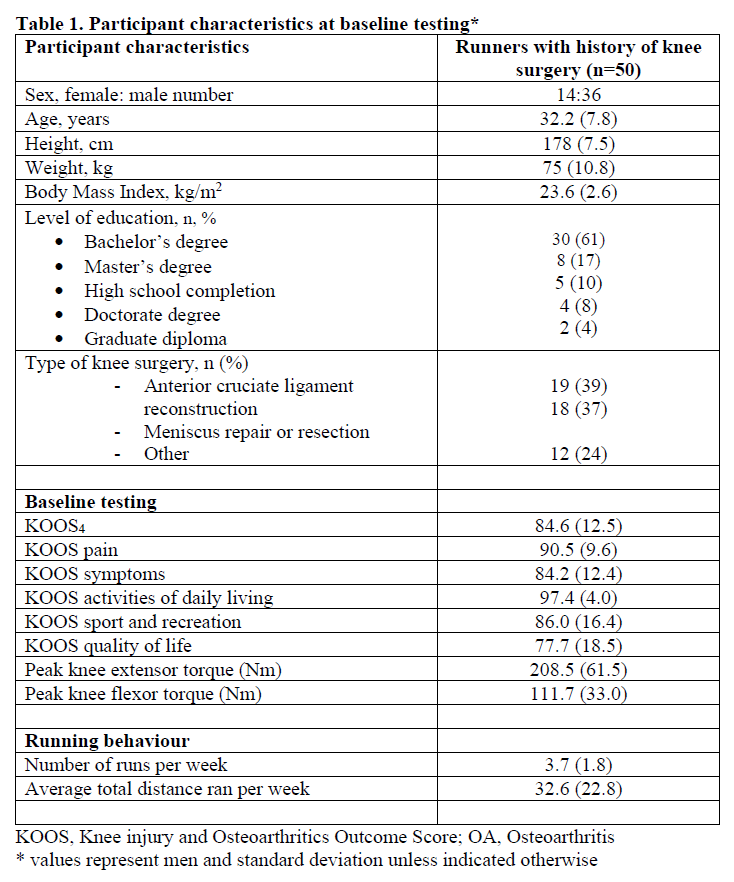
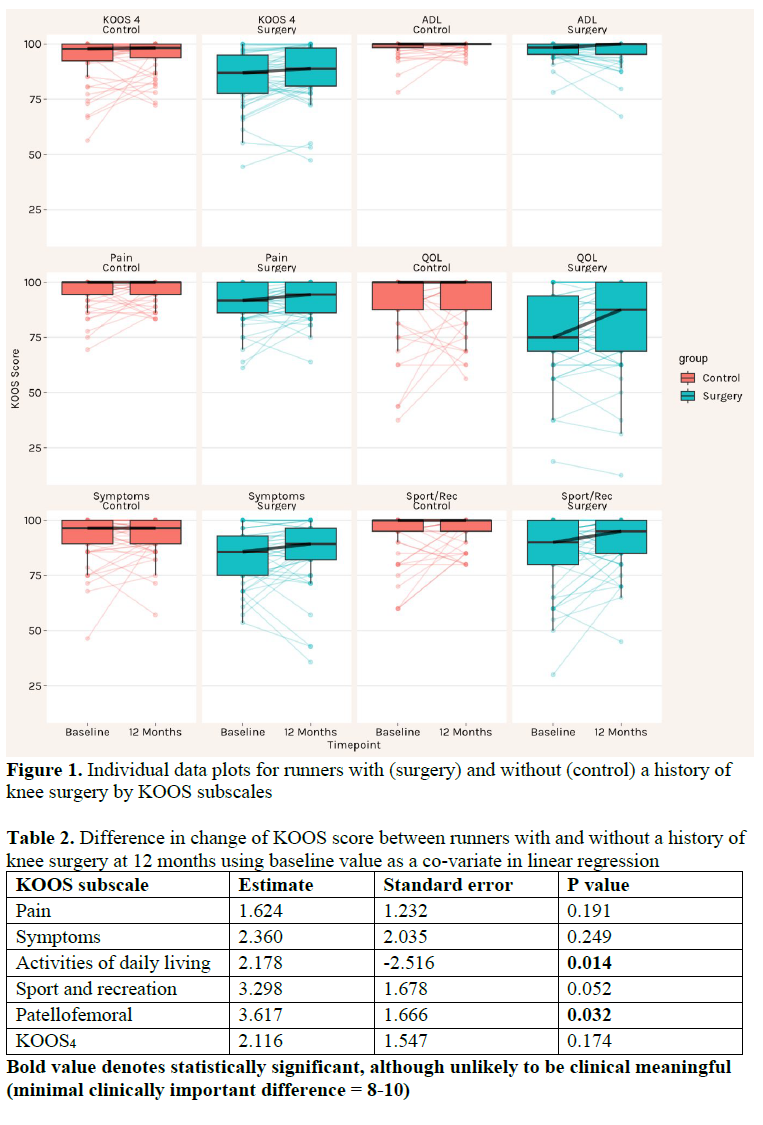
Once analysis of the MRI scans has been completed (end of 2023), we will explore the association of running behaviour on knee OA structural features using the MOAKS (MRI Osteoarthritis Knee Score) (8).
Grants
In 2021-2022, through a competitive process, we successfully obtained additional funding, >$60,000 across two La Trobe University grant schemes to: 1) increase recruitments numbers to reach 100 runners with, and 100 runners without a history of knee surgery, 2) recruit additional women runners to achieve sex and gender parity and explore women-specific factors (e.g., menstrual cycle, caring roles) on future knee OA outcomes, 3) long-term follow-up at 4 years, and 4) explore inflammatory blood biomarkers among high-risk runners.
1. Bruder AM, Crossley K, Mentiplay B, Culvenor AG, Johnston R, Hill A“Is running bad for your knees? The first study to explore whether running might NOT increase the risk of knee osteoarthritis in young adults.” La Trobe University Research Scheme. Awarded $45 685
2. Mentiplay B, Haberfield M, Briggs, C, De Oliveira Silva D, Bruder AM, Thornton J, Crossley K, Title: “Are biomechanical variables associated with future knee symptoms and injury in women runners at risk of early knee osteoarthritis?” La Trobe University Research Scheme. Awarded $24, 632
We have already commenced recruiting additional runners with a history of knee surgery to undergo physical assessments, MRIs, questionnaires, and blood collection. The increased sample size will allow us to perform sufficiently powered analyses of the preliminary findings enabled by this grant.
WHAT PROBLEMS, IF ANY, DID YOU ENCOUNTER?
At this stage we have not had the MRI images graded. To address this, eight early-career researchers have been trained by Professor Edwin Oei (experienced musculoskeletal radiologist, Erasmus MC, Netherlands) using MOAKS (MRI Osteoarthritis Knee Score) (8). This team of researchers will analyse the 50 MRIs to understand knee OA features in runners with a history of knee surgery. The final results will be available at the end of 2023.
HAVE YOU DISSEMINATED, OR PLAN TO DISSEMINATE, THE RESULTS OF THIS RESEARCH?
The final analysis of all MRI scans will be completed by researchers trained in semi-quantitative MRI evaluation of knee OA using the MRI OA Knee Score (MOAKS). This will be completed by the end of 2023. Thus, the major dissemination of the outcomes of this grant will occur once this analysis is completed. Following this, data will be written up in several planned publications for submission to high-impact peer-review international rheumatology journals such as Osteoarthritis and Cartilage, Arthritis Care and Research, and sports medicine journals such as British Journal of Sports Medicine. At this time point Arthritis Australia will be informed and given PDF copies of papers published.
We have published our larger study protocol, of which this study forms a critical part.
Reference
1. De Oliveira Silva D, Johnston RTR, Mentiplay BF, et al Trajectory of knee health in runners with and without heightened osteoarthritis risk: the TRAIL prospective cohort study protocol BMJ Open 2023;13:e068040. doi: 10.1136/bmjopen-2022-068040
The grant has enabled wider collaborations with international running researchers. Our research group is coordinating an international workshop on running and knee health alongside the World Congress of OA in Denver, USA on 17th March 2023. This workshop will include researchers from 5 difference research groups worldwide (U Kentucky & UCSF (USA), Uni BC (CAN), Erasmus (NETHERLANDS), La Trobe (AUS).
Conference presentations
Data collected from this grant has contributed to additional analyses and presented at two conferences:
1. Presenting Author: Danilo De Oliveira Silva. Presentation title: Runners with noisy knees after surgery: Is it related to symptoms? at Sports Medicine Australia, Brisbane, Australia, Nov 2022.
– This presentation was shortlisted for best paper award in clinical medicine stream.
2. Presenting Author: Mick Girdwood. Presentation title: Thigh muscle strength is not related to self-reported function in the medium to long term after anterior cruciate ligament reconstruction at Sports Medicine Australia, Brisbane, Australia Nov 2022.
Reference List
1. Culvenor AG, … Crossley KM. Is patellofemoral joint osteoarthritis an under-recognised outcome of anterior cruciate ligament reconstruction? A narrative literature review. Br J Sports Med. 2013;47(2):66-70.
2. Lohmander LS, et al. The long-term consequence of anterior cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med. 2007;35(10):1756-69.
3. Filbay SR, … Crossley KM. Health-related quality of life after anterior cruciate ligament reconstruction: a systematic review. Am J Sports Med. 2014;42(5):1247-55.
4. Esculier J-F, et al. What are the perceptions about running and knee joint health among the public and healthcare practitioners in Canada? PloS one. 2018;13(10).
5. Qiu L, et al. Biochemical changes in knee articular cartilage of novice half-marathon runners. 2019;47:5671-9.
6. Esculier J-F, et al. Cartilage recovery in runners with and without knee osteoarthritis. The Knee. 2019.
7. Øiestad BE, et al. Knee extensor muscle weakness is a risk factor for development of knee osteoarthritis. A systematic review and meta-analysis. Osteo Cart. 2015;23(2):171-7.
8. Hunter DJ, et al. Evolution of semi-quantitative whole joint assessment of knee OA: MOAKS. Osteo Cart. 2011;19(8):990-1002.
Targeting a novel pathogenic neutrophil subset in SLE.
| Funded by: | ARA Research Fund |
| Recipient: | Dr Katherine Martin and Professor Ian Wicks |
| Intended Department | WEHI – Walter and Eliza Hall Institute of Medical Research |
| Project: | Targeting a novel pathogenic neutrophil subset in SLE |
Scientific Report
Background
Systemic lupus erythematosus (SLE) is an autoimmune disease with a wide spectrum of clinical features. In this disease, chronic inflammation causes organ damage, with poor outcomes for patients. Despite decades of research, we still do not fully understand what causes this or how best to treat it. Furthermore, biomarkers that predict disease activity are severely lacking. While research has predominantly focused on T and B cells, it is becoming increasingly clear that neutrophils play an important role in SLE. While essential for host defence, dysregulated neutrophil responses contribute to the pathogenesis of SLE. Neutrophils become hyperactive in SLE, resulting in the release of proinflammatory molecules and mediators of tissue damage. Formation of neutrophil extracellular traps (NETs) is enhanced in SLE, leading to autoantigen exposure, and inciting the production of autoantibodies. NETs also directly promote tissue damage. While neutrophils were long considered a homogeneous population, it is now recognised that pathogenic subsets can appear in disease. Using single cell profiling, we have identified a novel neutrophil subset in SLE, which is absent or minimal in healthy people. This subset is defined by expression of CD98, an essential amino acid transporter. CD98 is responsible for importing essential amino acid into cells, which in turn fuels the mitochondria to produce energy. CD98 neutrophils are more metabolically active, which amplifies their pathogenic capacity. Our preliminary data suggests a correlation between CD98 neutrophils and disease activity in SLE, potentially providing a new biomarker.
Hypothesis
We hypothesize that CD98 neutrophils are pathogenic and targeting this subset may prevent neutrophil-mediated inflammation and organ damage in SLE. We also hypothesize that the CD98 neutrophil population represents a novel clinical biomarker for SLE.
Grant objectives and aims:
Our overall goal was to decipher the role of CD98 neutrophils in SLE. In this project funded by
Arthritis Australis, we addressed the following aims:
Aim 1: Validate CD98 neutrophils as a novel biomarker in SLE.
Aim 2: Target CD98 on neutrophils to limit disease in a preclinical model of SLE.
Results
Aim 1: LDN are expanded in PB of SLE patients and are pathogenic.
We sought to determine if CD98 neutrophils could be used a prognostic biomarker to indicate disease activity in SLE. For this study, patients with active disease were recruited through the Rheumatology Unit at the Royal Melbourne Hospital and healthy control donors were recruited through the Volunteer Blood Donor Registry at WEHI. Peripheral blood was collected and the number of CD98 neutrophils was quantified using flow cytometry. Very few CD98 neutrophils were detected in the blood of healthy individuals, while SLE patients had increased numbers of this subset. Strikingly, the number of CD98 neutrophils in peripheral blood of SLE correlated with disease activity scores as measured by SLEDAI-2K. This suggests that quantification of CD98 neutrophils in peripheral blood could serve as a novel prognostic biomarker to measure disease activity, remission or response to treatment and assist with clinical management of SLE patients.
We conducted further analysis of the CD98 neutrophils compared to normal neutrophils that do not express CD98. We assessed size, granularity, and the maturation status of the two neutrophil populations in SLE using flow cytometry. CD98 neutrophils were comparable in size, but more granular than normal neutrophils, as indicated by an increase in mean side scater. This is in keeping with our observation that CD98 neutrophils express more granule proteins including elastase, proteinase 3 and myeloperoxidase, all known to cause endothelial and tissue damage in autoimmune diseases. CD98 neutrophils were comprised of both mature and immature neutrophils, as determined by CD10 expression. Functionally, CD98 neutrophils were resistant to cell death and capable of generating more proinflammatory cytokines and chemokines, especially IL6, with a 40-fold increase above baseline. Lastly, our results suggest that the pathogenic capacity of this neutrophil subset may be directly due the expression of CD98. CD98 neutrophils were more energetic and produced energy through both glycolytic and mitochondrial dependent pathways, whereas normal neutrophils generate energy through glycolysis only. Using a potent CD98 inhibitor (JPH203), we demonstrated that CD98 is directly responsible for fuelling the increased bioenergetic capacity in this neutrophil subset (Fig 2D). In these experiments, JPH203 inhibited CD98 mediated accumulation of amino acids that are essential for powering mitochondrial energy production, which in turn may facilitate their proinflammatory function.
Aim 2: CD98 neutrophils are present in a highly relevant animal model of SLE.
To evaluate whether CD98 neutrophils contribute to the pathogenesis of SLE in vivo, we used the Lyn-deficient (Lyn-/-) mouse model. This is a well described experimental model of SLE that develops the serological and pathological hallmarks of the disease including autoantibody production, inflammation, and glomerulonephritis. Activation of the autoimmune response can be observed as early as 12 weeks and mice develop severe disease and renal damage by 36 weeks. Given CD98 neutrophils are found in SLE patients but not healthy people, we first evaluated levels of this neutrophil subset in Lyn-/- mice. Cohorts of Lyn-/- mice with mild (12 weeks), moderate (24 weeks) or severe (36 weeks) disease were evaluated. Age matched wild type control mice were also assessed at each timepoint. While CD98 neutrophils were present at similar levels in the bone marrow of wild type and Lyn-/- mice at all timepoints examined, wild type control mice had very few CD98 neutrophils within the spleen or peripheral blood, regardless of age. Compared to 12 week old control mice, Lyn-/- mice with mild disease had a small increase in CD98 neutrophils in the periphery. Importantly, similar to human disease, Lyn-/- mice exhibiting moderate to severe disease had a markedly increased number of CD98 neutrophils in their blood and spleen compared to Lyn-/- mice with mild disease. This indicates that CD98 neutrophils are behaving similarly in the Lyn-/- mouse model to human SLE patients. We believe the Lyn-/- mice therefore represent a useful model to understand how the CD98 neutrophil subset contributes to SLE.
Ongoing research
We have strong evidence to show that CD98 neutrophils are pathogenic and may contribute to inflammation and tissue damage in SLE. We also have in vitro data to show that targeting CD98 may limit their pathogenic capacity. To evaluate this directly, we will specifically delete CD98 from all granulocytes and granulocyte progenitors by crossing the CD98flox mice with the neutrophil conditional MRP8Cre mice. Using funds provided by Arthritis Australia, we have purchased CD98flox mice from Jackson laboratories and established a colony at WEHI. MRP8Cre mice are available in-house at WEHI and we have commenced breeding a CD98 conditional neutrophil knockout mouse. Ongoing research continuing in 2023 will generate Lyn-/- mice lacking CD98 in this neutrophil subset to determine whether targeting CD98 can limit neutrophil-mediated inflammation and tissue damage and thereby decrease the severity of SLE.
Grant outputs
Publications
Martin KR, Day JA, Hansen JA, D’Silva DB, Wong HL, Garnham A, Sandow JJ, Nijagal B, Wilson N, Wicks IP. (2023) CD98 defines a metabolically flexible, proinflammatory subset of low- density neutrophils in systemic lupus erythematosus. Clin Transl Med. 2023 Jan:13(1)
Conference presentations
- Oral presentation, Society for Leukocyte Biology Annual Meeting 2022 (Hawaii, USA). CD98 defines a metabolically flexible, proinflammatory subset of low-density
- Oral presentation, Australian Inflammation Centers 2022 Symposium (Melbourne). CD98
- a novel regulator of inflammation in rheumatoid arthritis
- Oral presentation, Australian Rheumatology Association (ARA) Annual Scientific Meeting 2021 (Virtual). Identification of a novel pathogenic neutrophil subset in SLE.
Plain language summary
What is the problem?
Systemic lupus erythematosus (lupus) is an autoimmune disease where the body begins to atack itself. People with lupus develop severe inflammation and tissue damage that affects many different organs and tissues throughout the body including the skin, joints, heart, kidney, nerves, and blood vessels. Over 20,000 Australians and 5 million people worldwide are estimated to be living with this disease. Despite recent advances in our understanding of what causes lupus and some improvement to available treatments, the prognosis for many patients remains poor and quality of life is severely impacted. Treatments for this disease can be ineffective, non-specific, have many serious side effects and can leave patients susceptible to life threatening infections. In fact, only two new drugs have been approved for lupus in the past 20 years and these new treatments unfortunately have limited use in the clinic as only a very small group of patients benefit from their use. Therefore, there is a need to identify new causes of this disease so we can develop new and beter therapies to treat lupus.
Identification of a new population of disease-causing immune cells in lupus
One type of white blood cells thought to contribute to the inflammation and organ damage in lupus are neutrophils. Neutrophils are the most abundant immune cells in the blood and play an essential role in protecting the body against infection. On the other hand, hyperactive neutrophils can cause chronic inflammation and tissue damage in a range of autoimmune diseases. Indeed, recent research in both human patients and animal models of lupus have shown that neutrophils can release toxic molecules and signals that cause inflammation, organ, and tissue damage, all of which are associated with lupus. Importantly, while neutrophils were long considered to be a homogenous population of cells, there is compelling evidence that certain subsets of neutrophils are disease causing and can promote inflammation and tissue damage in human disease but are absent in healthy people.
Using advanced technologies, we were the first to identify a new neutrophil population that is present in the blood of lupus patients with active disease. These neutrophils can be identified by a protein on their cell surface called CD98. CD98 can transport a type of fuel called amino acids into cells, and these amino acids are then used by cells to generate energy.
Our idea
We propose that CD98 neutrophils cause inflammation and tissue damage in lupus and that targeting these disease-causing neutrophils could be a new way to treat this debilitating autoimmune disease. We will address our aims using blood collected from human lupus patient as well as from mouse models of lupus.
What did we discover?
Biomarkers are something that doctors can assess in patients that tells them how severe disease is, whether treatments are working, whether patients are in remission or are having a flare of symptoms. Biomarkers in lupus are severely lacking and this hinders the way doctors can properly manage these patients and impedes development of new therapeutics. Using funding from Arthritis Australia, we collected blood from human lupus patients and showed that CD98 neutrophil levels correlated with “disease activity” (Martin et al, 2023, Clinical and Translational Medicine). This means that patients with severe lupus symptoms had more of these neutrophils in the blood when compared to patients with mild disease and this population was completely undetectable in healthy people. This exciting finding suggests that the levels CD98 neutrophils could represent a “biomarker” to be used by doctors as a simple and effective test to help guide patient care or to assess whether treatments are working. In addition to showing that CD98 neutrophils correlate with the severity of lupus, we also show that they can cause an inflammatory response in patients. For example, we showed that CD98 neutrophils produce proinflammatory signals, release toxic molecules that cause cellular and tissue damage and are more metabolically active which enhances their ability to cause damage.
To understand exactly how CD98 neutrophils contribute to lupus and to determine if targeting this population could be new way to treat this autoimmune disease, we turned to a mouse model that develops many of the same clinical features as human lupus. These mice develop systemic inflammation, produce of self-targeting proteins called autoantibodies and show damage to organs such as the kidney. These mice develop mild lupus symptoms at 24 weeks of age and show severe signs of disease at 36 weeks of age. Given that in human disease, CD98 neutrophils are found in patients with lupus but not in healthy people, we asked the question of whether these cells were also found in the mouse model of lupus. We looked at mice with no disease symptoms (12 weeks of age), mild (24 weeks of age) or severe symptoms (36 weeks of age) and compared them to normal mice. In these experiments, we found that before disease developed (12 weeks), the lupus mice had equivalent levels of CD98 neutrophils as normal healthy mice. Similar to human patients, lupus mice with mild or severe disease symptoms had a large increase in the number of CD98 neutrophils in their blood compared to normal healthy mice. This indicated that CD98 neutrophils are behaving similarly in the lupus mouse model as they are in human patients and means we can use these mice beter understand the role these neutrophils playing in disease and find ways to target them and limit their disease causing capacity.
Where to next?
We have strong evidence to show that CD98 neutrophils contribute to lupus. We also have exciting data to show that if we target CD98 on this neutrophil population in a test tube we can limit the amount of damage they can cause. The next step of this project is to determine if targeting CD98 on the disease causing neutrophil population can prevent or treat lupus. To do this, we have used funds from Arthritis Australia to purchase genetically modified mice that do not have CD98 on the surface of the neutrophils. Our next step is to breed these mice with the lupus mice and determine whether absence of CD98 can limit the damage these neutrophils can cause and decrease the severity of lupus symptoms.
How will this benefit people with lupus?
Biomarkers represent an essential part of clinical practice and are used in almost every step of patient care. Biomarkers that predict disease outcomes, remission, and long-term adverse events in lupus are severely lacking and this hinders the way doctors can properly manage these patients and impedes development of new therapeutics. We have developed an easy and simply test clinicians can use to guide the way they treat patients. Furthermore, we are providing the proof-of-concept evidence that CD98 on these disease causing neutrophils represents an exciting new therapeutic target to treat lupus.
Unravelling Metabolic Mechanisms in Knee Osteoarthritis Pain.
| Funded by: | Arthritis Australia and Suzette Gately |
| Recipient: | Dr Feng Pan |
| Intended Department | Menzies Institute for Medical Research, University of Tasmania |
| Project: | Unravelling Metabolic Mechanisms in Knee Osteoarthritis Pain |
Plain language summary (Lay report)
Give a brief overview of the research you have undertaken, what you hoped to achieve and what you have achieved.
Osteoarthritis (OA) is a common and painful condition that affects many people in Australia. It’s a disease that gets worse over time and there is no cure for it. The main symptom of OA is pain, which is why people seek medical help. Unfortunately, the current treatments for OA only manage the pain and are not very effective. This is because we don’t fully understand why people experience pain differently and why the treatments don’t work for everyone. We need better ways to understand and treat pain in OA, which is why my research is focused on understanding the different types of pain people experience and how we can develop better treatments that are tailored to the individual. By doing this, we can improve the lives of people living with OA and provide better care for them.
My research has shown that there are different types of pain in OA, and that these types can be split into distinct groups. These groups have different underlying causes and can affect people in different ways. By understanding the different groups, we can better treat the pain and improve people’s lives. However, we still need to learn more about the different types of pain and develop better ways to identify them in order to provide tailored treatments that work for each individual.
What questions did the grant set out to answer? What problems did you try to solve, or gaps in knowledge did you try to fill? Why is this important?
Recent research suggests that people with metabolic problems may be at higher risk of developing OA and experiencing more severe pain. Metabolomics is a new technology that can help identify small molecules in the body that are related to diseases like OA. By using targeted serum metabolic profiling, we’ve found a lipid metabolite (sphingomyelin (SM) C18:1) that’s linked to multisite pain in people with OA. This suggests that lipid metabolism may play an important role in causing pain in OA. Currently, no studies have identified metabolites specifically linked to OA-related pain and its evolution. Our project aims to find more biomarkers that are linked to different types of OA pain in order to develop better ways to diagnose and treat this condition based on untargeted metabolomics platform. The use of metabolomics technology will help us find specific signs in the body (called biomarkers) that can help doctors figure out which treatment will work best for each patient. It may also lead to the development of new treatments for osteoarthritis pain.
What did you discover during the course of the grant?
This grant enabled us to measure serum metabolites using metabolomics in our cohort. We measured 536 and 335 samples from Phase 2 and Phase 4 of cohort, respectively.
The project looked at the chemicals in the body that are related to pain in people with osteoarthritis (OA). We found that certain types of fats (lipids) were associated with pain at different phases of the study, even after taking into account things like age, sex, body weight, physical activity, and diabetes. In total, we found 154 different types of lipids that were linked to pain at different times throughout the study. Some of these lipids were associated with pain both in the short term and over time, suggesting that the way the body handles fats may play a role in how much pain someone with OA experiences.
The project also found that certain types of lipids, which are molecules that are involved in metabolic processes in the body, are associated with bone health. In particular, we looked at bone mineral density (BMD) and fracture risk. We found that certain lipid species and classes were associated with hip BMD at different time points throughout the study, and that most of these associations persisted over time. Additionally, We identified certain lipids that were associated with an increased risk of fracture, both at the beginning of the study and over time. Interestingly, some of these associations were independent of BMD, which suggests that lipids may play a direct role in bone health.
Overall, this project provides new insights into the complex relationship between lipids, pain and bone health. By identifying specific lipids that are associated with pain, BMD and fracture risk, we have highlighted potential targets for future interventions aimed at reducing pain and improving bone health. Additionally, our findings could have important implications for the development of personalised medicine approaches for the prevention and treatment of pain, osteoporosis and other bone-related conditions.
Have the findings of the research already benefitted people with musculoskeletal disease? How might the findings inform further research to help sufferers in the future?
Although our project grant’s research findings have not directly benefitted individuals with OA, our metabolomics research has shown promise in enhancing our knowledge of pain and bone mineral density (BMD) and identifying potential new therapeutic targets. Our findings may aid researchers in comprehending the underlying mechanisms of pain and BMD, leading to more effective pain and osteoporosis treatments. The identification of metabolites involved in pain and bone metabolism pathways may also allow researchers to develop drugs that target these metabolites, reducing pain and bone loss.
Are you planning to continue the research?
Yes, we have identified some metabolites that are involved in pain and bone metabolism pathways, indicating their biological relevance to both pain and bone mineral density (BMD). To confirm our findings, we plan to replicate them in external cohorts. Additionally, we intend to apply for NHMRC project grants based on these promising preliminary results.
Finally, I would like to acknowledge the Arthritis Foundation of Australia and Suzette Gately for making this research possible. I want to take this opportunity to express my heartfelt gratitude for your generous donation towards Arthritis Australia Project Grant. Your support and generosity have allowed us to continue our mission of better pain treatment and improving patients’ quality of life, and we would not have achieved this without your support.
Dr Feng Pan (Menzies Institute for Medical Research, University of Tasmania)
Scientific summary (Scientific report)
What were the main scientific objectives of the grant?
The main objective of this project aimed to identify and validate biomarkers with different OA pain presentations using untargeted metabolomics platform.
What were the main scientific achievements of the grant?
By analysing the metabolomics data, we found at Phase 2, three lipid species of TG(O-52:0)[SIM], PE(36:0) and PE(15-MHDA_22:6) and class of FFA were associated with pain score after controlling for age, sex, body mass index, physical activity and diabetes.
Four lipid species of PC(O-18:0/18:2, 34:2), PC(42:5)(a) and Hex2Cer(d16:1/24:1) and class of Hex2Cer were associated with pain score at the phase 4 after controlling the same covariates. There were 154 lipid species and 21 lipid classes associated with pain score from Phase 2- to Phase 4. Of them, 23 interaction terms of and follow-up times and 10 interaction terms of lipid classes and follow-up times were associated with pain score from Phase 2- to Phase 4. Lipid specie of PE(36:0) and class of FFA were associated with pain score cross-sectionally and longitudinally. These results suggest that lipid dysregulation has a role in OA pain severity and change over time.
Other than these findings, we also identified lipid metabolic markers of bone mineral density (BMD) and fracture given that recent studies suggest that lipids play a role in bone metabolism. However, the relationship between lipids and BMD using untargeted metabolomics profiling has not been explored.
At Phase 2, there were 7 lipid species of phosphatidylethanolamine (PE)(16:0/20:5, 16:0/22:6, 18:0/22:6, 38:5(b)), triacylglycerol (TG)(50:0)[ (neutral loss (NL)-18:0], TG(54:6)[NL-22:6], phosphatidylinositol (PI)(34:0) and 3 classes of PE, phosphatidylglycerol (PG), alkyldiacylglycerol (TGO) associated with hip BMD after controlling covariates. At Phase 4, there were 9 lipid species of PC(O-18:1/18:1, 18:0/18:1), lysoalkylphosphatidylcholine (LPC(O)-22:1), alkenylphosphatidylethanolamine (PC(P)-18:1/18:1), Hex2Cer(d18:2/24:1, 16:1/24:1, 18:1/16:0), trihexosylcermide (Hex3Cer)(d18:1/16:0), sphingomyelin (SM)(d18:0/16:0) and 5 classes of Monohexosylceramide (HexCer), Hex2Cer, Hex3Cer, LPCO, GM3 associated with hip BMD after controlling covariates. There were 192 lipid species and 27 lipid classes associated with hip BMD from phase 2 to phase 4. Most of interaction terms of these lipid species and classes with follow-up times were associated with hip BMD from phase 2 to phase 4. Of them, lipid species of PE(38:5) (b), PI(34:0), Hex2Cer(d16:1/24:1, d18:1/16:0, d18:2/24:1), TG(50:0)[NL-18:0], TG(54:6)[NL-22:6], PC(P-18:1/18:1), PC(O-18:1/18:1, 18:0/18:1), SM(d18:0/16:0), classes of PE, PG, HexCer, Hex2Cer, Hex3Cer, TG(O), GM3, LPCO were associated with hip BMD cross-sectionally and longitudinally.
At Phase 2, sphingosine-1-phosphate (S1P)(d16:1) and Hex2Cer(d18:2/16:0) were associated with the prevalence of fracture in univariable and multivariable analyses. Furthermore, S1P(d16:1) was associated with fracture independent of hip BMD at Phase 2. However, there were no lipid classes associated with fracture at Phase 2. At Phase 4, deoxyceramide (Cer)(m18:0/20:0), phosphatidylcholine (PC)(14:0/20:4) and TG(50:4)[NL-20:4], TG(54:1)[NL-18:1] and LPC(O-18:1) were associated with fracture after controlling covariates and hip BMD. Three lipid classes of LPC(O), hydroxylated acylcarnitine (AC-OH) and PE were associated with fracture at Phase 4 after controlling covariates and hip BMD. There were 5 lipid species of Cer(d17:1/16:0), Hex2Cer(d16:1/24:1, 18:2/16:0, 18:2/24:1), acylcarnitine (AC)(24:1)(a) and 4 lipid classes of PS, Hex2Cer, Hex3Cer and GD1 ganglioside (GD1) were associated with incident fracture from Phase 2 to Phase 4.
What problems, if any, did you encounter in achieving the project’s objectives, and how did you address them?
The problem we encountered in achieving this project’s objective was the complexity of the data analysis process. We addressed this problem by collaborating with researchers from the Barker Institute in statistical analysis and bioinformatics, using advanced software and computational tools.
Have you disseminated, or plan to disseminate, the results of this research?
We have one paper published (Ma C, Liu M, Tian J, Zhai G, Cicuttini F, Schooneveldt YL, Meikle PJ, Jones G, Pan F. Lipidomic Profiling Identifies Serum Lipids Associated with Persistent Multisite Musculoskeletal Pain. Metabolites. 2022 Feb 25;12(3):206. doi: 10.3390/metabo12030206. PMID: 35323649; PMCID: PMC8953175.)
We are now writing up the second paper about the lipids markers and pain severity across Phase 2 to Phase 4.
We also working on a paper on lipids and BMD and incident fracture.
Papers that have been submitted and/or accepted for publication
Ma C, Liu M, Tian J, Zhai G, Cicuttini F, Schooneveldt YL, Meikle PJ, Jones G, Pan F. Lipidomic Profiling Identifies Serum Lipids Associated with Persistent Multisite Musculoskeletal Pain. Metabolites. 2022 Feb 25;12(3):206. doi: 10.3390/metabo12030206. PMID: 35323649; PMCID: PMC8953175.)
Meetings/conferences at which you have presented this research, or are due to present it (please provide abstracts if possible)
The work about lipids with multisite musculoskeletal pain has been presented at the 2022 EULAR and WCO-IOF-ESCEO conferences.
Finally, I would like to acknowledge the Arthritis Foundation of Australia and Suzette Gately for making this research possible. I want to take this opportunity to express my heartfelt gratitude for your generous donation towards Arthritis Australia Project Grant. Your support and generosity have allowed us to continue our mission of better pain treatment and improving patients’ quality of life, and we would not have achieved this without your support.
Dr Feng Pan
Menzies Institute for Medical Research, University of Tasmania
Creaky knees and exercise beliefs and participation. A mixed-methods study.
| Funded by: | ARA Research Fund |
| Recipient: | Jeanette Thom |
| Intended Department | School of Health Sciences, UNSW Sydney |
| Project: | Creaky knees and exercise beliefs and participation. A mixed-methods study. |
Plain language summary:
This report entails the planning, methodology, results and subsequent actions of our interview study into the influence that knee crepitus has on peoples’ emotional and physical behaviours. Specifically, we sought to discover if people have concerns about knee crepitus, and whether these concerns influence decisions regarding exercise.
Crepitus: We have defined crepitus as any noise or sound that people may perceive coming from their knee joints.
BACKGROUND: A literature review of the current information about osteoarthritis, crepitus and patient perspectives/attitudes towards crepitus and exercise in general confirmed several things:
- Osteoarthritis is a significant burden of disease in Australia.
- Crepitus can be defined as a range of sounds elicited from any joint, which can sometimes be palpable.
- Crepitus can be present in disease and normal states and may not be regularly measured for diagnostic purposes.
- Although clinicians and health professionals place little emphasis on the presence of crepitus, it is something that can cause concern for patients.
- Exercise is effective for prevention and treatment of joint health, however, many Australians do not meet current Australian exercise guidelines.
WHAT WE DID: Therefore, we conducted focus group and individual interviews of people with knee crepitus to inform the creation of a wider survey to collect information about the perspectives and exercise behaviours of people with crepitus. We theorised that people with knee crepitus may be concerned about their knees and hesitant or worried about exercise/movement that elicited any sounds.
After receiving ethics approval from UNSW Sydney, we commenced recruitment for the study. Recruitment was conducted through email invitations, social media posts and flyers in public places (e.g. doctor’s office, cafes, gyms). People included were anyone aged over 18 years who had knee crepitus, with or without osteoarthritis, and with some level of concern about their crepitus. Twenty-four participants consented to take part in the study. The participants ranged in age from 21-69 years.
Two researchers guided the interviews, in a way that allowed for stimulation of conversation and thorough history taking of the participants’ knee crepitus and experiences. Transcripts from these interviews were then analysed.
WHAT WE FOUND: Five key themes emerged, guided by the questions asked of the participants. These were:
- the types of crepitus that the participants described varied
- when they experienced their crepitus
- what their crepitus meant to them
- their attitudes and exercise behaviours regarding crepitus
- the sources of advice received or wanted concerning their crepitus.
The results revealed that crepitus came in a range of different sounds for participants and could be variable depending on types of movement. Whilst no distinct patterns could be identified between types of movement and types of sounds, a range of descriptions were provided, for example from a “squeak” to the “loudest creak she’d ever heard”, and ranged in description, such as “squeak”, “little pops”, “click”, “grating”, “cracking” and sounding like a “tin man”.
The presence of crepitus caused a level of concern in most participants. However, it was not a high priority concern for most and their true concerns were only elicited from deeper questioning. For example, a participant described initially not being too concerned about the noise, but later admitting that they were worried it meant they would need a joint replacement sooner. Participants who had associated symptoms, particularly pain, stiffness, locking or catching, were more concerned about other symptoms than their crepitus.
Most participants were physically active and wanted to remain active. They all thought that exercise was good for their knee joint health, though it may depend on the type of exercise. The ‘right type’ of exercise that was considered beneficial for joints varied. Though most believed that strengthening exercises and non-twisting/lower impact exercises were positive for their joints. Generally, participants had not ceased exercise, but they may have modified their activities due to crepitus and associated symptoms. Some people had intentionally done more strength training to try alleviating their crepitus.
Participants agreed that more understanding about what causes crepitus and what exercise was safe for knee health would be beneficial. People with crepitus are interested to understand more about the cause of crepitus, and to be guided by health professionals as to what they can do to remain physically active, and if appropriate delay the onset of more concerning symptoms, such as pain.
HOW WE WILL USE THESE FINDINGS TO BENEFIT PEOPLE / INFORM FURTHER RESEARCH TO HELP PEOPLE IN THE FUTURE:
This study has informed researchers’ understanding of the attitudes and beliefs of people with knee crepitus, allowing them to be better equipped to conduct broader study on the topic. With the current findings, we plan to conduct a larger population survey to capture information we may have missed. This may include getting responses from people with a greater variety of exercise habits and knowledge about their own health. The survey will hopefully reinforce our current evidence for the attitudes and beliefs of people in the community with knee crepitus. The findings from these studies will be able to inform health professional approaches to education and management for people with crepitus and recognise opportunities to address any concerns, gaps in knowledge or misconceptions people may have. It is clear that people wish to be informed on what is going on in their own joints and want to be assured by health professionals what exercise is safe to do. From what we have learnt, we are now better equipped to do this.
WHY THIS PROJECT IS IMPORTANT:
Many people hear noises (crepitus) in their knees when they move. This can occur in people with osteoarthritis and those that do not have any other symptoms or conditions. It was important to find out how concerned people are when they hear crepitus in their knees and what they do about it. We found that people do not seem to be too concerned if they get crepitus with no other symptoms such as pain and they do like to keep active. Though they may modify what activities that they do. They also do not see a health professional to just to ask about their crepitus. This is important to know, as we can then try to find ways to promote positive messages to keep people active for their knee and overall health.
Scientific report:
MAIN SCIENTIFIC OBJECTIVES OF THE PROJECT:
The overarching aim of this project was to determine if the experience of having ‘noisy knees’ influences peoples’ beliefs and exercise behaviours prior to being diagnosed with osteoarthritis.
The contribution of this study to scientific knowledge is that it provides insight for researchers and medical professionals on the concerns and beliefs of people with knee crepitus, allowing for more evidence-based approaches to patient priorities and education.
MAIN SCIENTIFIC RESULTS OF THE PROJECT:
Outcomes to date:
The initial focus group study of this project has been completed, with 24 people affected by crepitus recruited for the online interviews / focus groups. The preliminary results from this study have been presented in a variety of settings (see below) and a manuscript is being finalised. These results are currently being used to inform a wider online survey study (Part B).
Key findings include:
- Five main themes emerged from the 24 participants:
- Individual variation of knee crepitus
- Occurrence of knee crepitus
- Meaning of knee crepitus
- Attitudes and exercise behaviours regarding crepitus
- Knowledge deficits and needs concerning crepitus
- Most participants had not ceased exercise but may have modified movement due to crepitus and associated symptoms, some had increased intentional strength training to try alleviating it.
- For those already with osteoarthritis or other symptoms, crepitus was of less concern than symptoms such as pain.
- Participants agreed that more understanding about the processes causing crepitus and what exercise was safe for knee health would be beneficial.
MAIN SCIENTIFIC ACHIEVEMENTS OF THE PROJECT:
UNSW Medicine Honours Thesis and presentation:
Patient perspectives on their creaky knees and the role of exercise in knee health: a qualitative study
Elise Drum, UNSW Medicine ILP Research Project, September 2022
CEPA: Clinical Exercise Physiology Association 2023 Virtual Conference presentation and published abstract:
Thom J., Kovats A., Drum E., Jones M., Dennis S., Mills K., & Naylor J. Patient perspectives on their creaky knees and the role of exercise in knee health. Journal of Clinical Exercise Physiology (2023) 12 (s1): 12. https://doi.org/10.31189/2165-7629-12-s1.12
There have also been presentations to community groups for potential recruitment, education and dissemination of the results. These include groups such as Arthritis NSW – Support Group, Windsor Bowls Team, clinical/research groups such as the Sydney NeuroMusculoskeletal Research Collaborative, Sydney Musculoskeletal Health -arthritis research theme, Riverina Orthopaedics (Wagga Wagga clinic) and within a podcast.
PLANS FOR FURTHER DISSEMINATION AND RESEARCH:
A manuscript of the focus group results is planned to be submitted for publication in 2023. Once this has been accepted for publication, further details will be provided and further dissemination via social media, community groups and researchers will take place.
Currently these results are informing Part B of the project, with the participants’ findings being used to develop the survey questions. The survey, following ethical approval, will be advertised for participants during 2023 and results disseminated later in early 2024.
Sign up to Arthritis Insights
Regular updates, news and research findings delivered to your inbox: